Regulatory News
Latest updates from regulatory authorities worldwide
🎤 Meet Our Speaker TraceHub Global Virtual Community Meeting
I'm honored to welcome Mr. Mohamed Youssef Chief Revenue Officer CRO of Juleb - AI ERP for Pharma Supply Chain Joining u...
🎤 Meet Our Speaker TraceHub Global Virtual Community Meeting
I'm thrilled, and proud to announce participation of EVOTEQ the leading force behind #Tatmeen (the national track and tr...
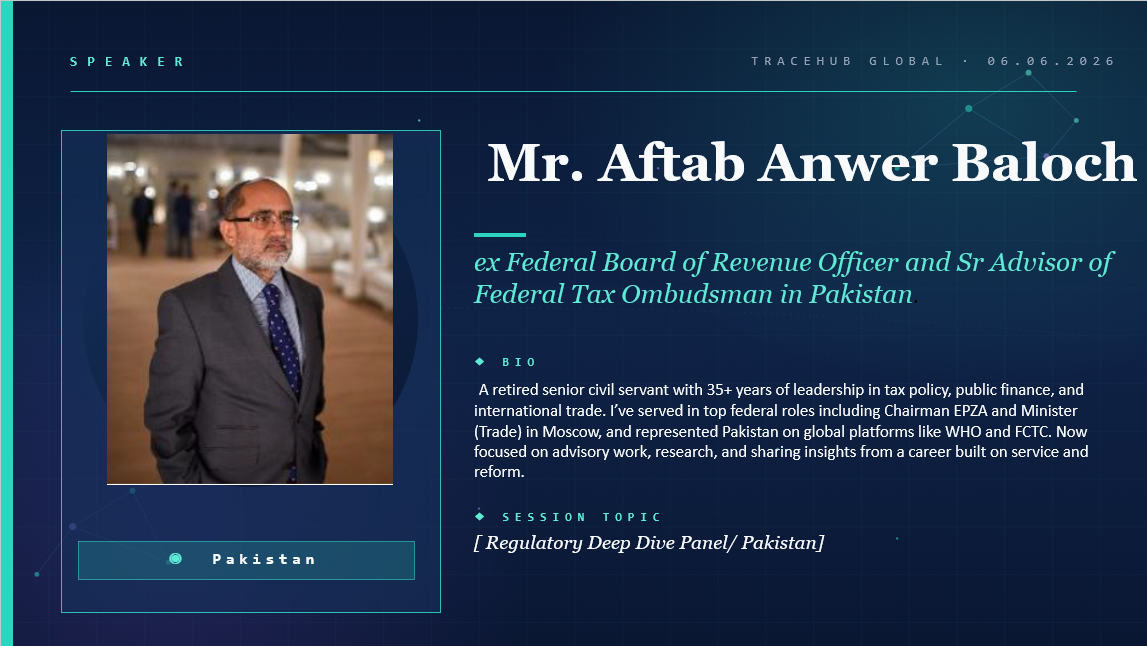
🎤 Meet Our Speaker TraceHub Global Virtual Community Meeting
Thrilled to annonce that TraceHub Global Virtual Community Meeting rringing together speakers and experts from 15+ count...
🎤 Meet Our Speaker TraceHub Global Virtual Community Meeting
I'm honored to welcome Mr. Pramod Gokhale , Sr President, Global CIO. Joining us at the 1st Ever Global Track & Trac...
🎤 Meet Our Speaker TraceHub Global Virtual Community Meeting
I'm honored to welcome Mr. Rizwan Buttar , Founder & Chief Innovation Officer, Pharma Trax Joining us at the 1st Eve...
🎤 Meet Our Speaker TraceHub Global Virtual Community Meeting
I'm honored to welcome Mr. Yousuf Jamjoom Founder and CEO of Juleb - AI ERP for Pharma Supply Chain Joining us at the 1s...
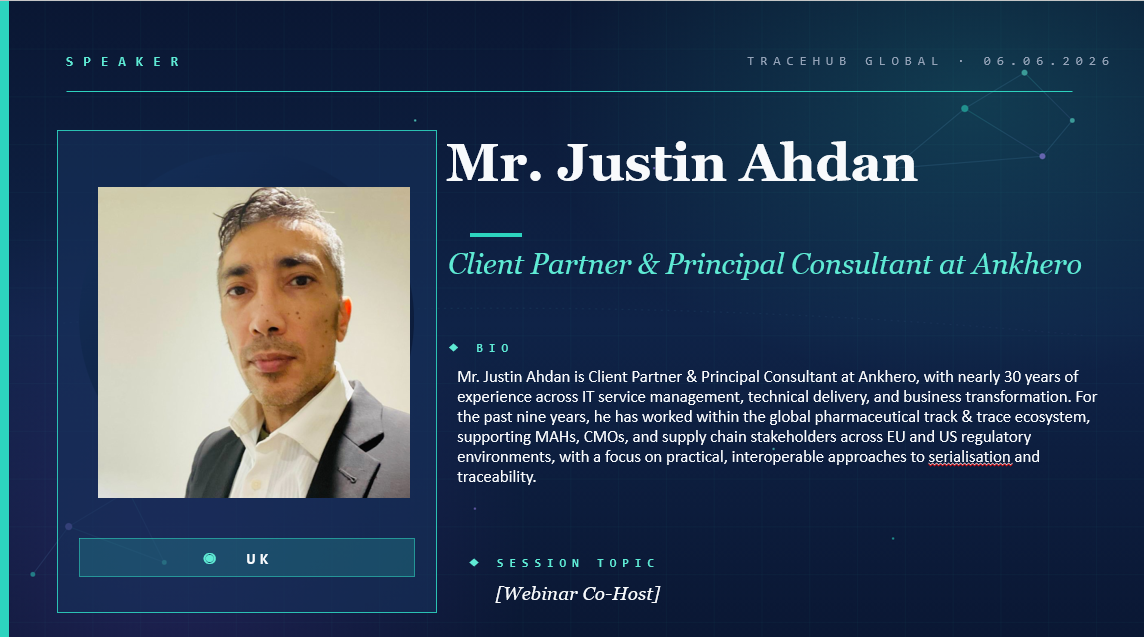
🎤 Meet Our Co-Host TraceHub Global Virtual Community Meeting
I'm honored to welcome my dear friend Mr. Justin Ahdan Client Partner & Principal Consultant at ANKHERO to co-host w...
🎤 Meet Our Speaker TraceHub Global Virtual Community Meeting
I'm honored to welcome Mr. Nathan Seyoum External Primary Medicine Registration Assessor Ethiopian Food and Drug Authori...
🎤 Meet Our Speaker TraceHub Global Virtual Community Meeting
I'm thrilled, and it is a personal honor for me to announce that Mr. Mehdi Amrouni ALGERIE MIPP will be a speaker at the...
🎤 Meet Our Speaker TraceHub Global Virtual Community Meeting
I'm honored to welcome Mr. Siddharth Reddy Co-Founder of AltiusHub Joining us at the 1st Ever Global Track & Trace C...
🎤 Meet Our Speaker TraceHub Global Virtual Community Meeting
I'm honored to welcome Mr. Patrick Maher , Independent pharmaceutical serialization and traceability consultant. Joining...
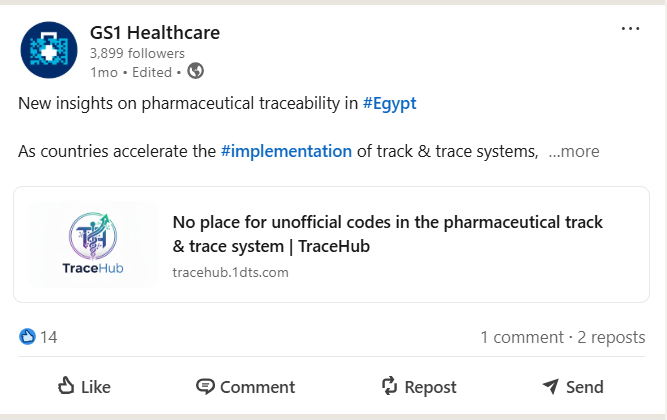
GS1 Healthcare Shares Information Published by TraceHub
A proud moment for TraceHub. Seeing GS1 Healthcare share and reference information published by TraceHub reflects the gr...