Knowledge Base for serialization engineers.
Learn about EPCIS, GS1 Standards, and Track & Trace implementation
General (28 articles)
A Reading of the Key Differences Between the Draft Regulatory Guide and EDA Law No. 804
There are several fundamental differences between the Draft Regulatory Guide governing the establishment and operation o...
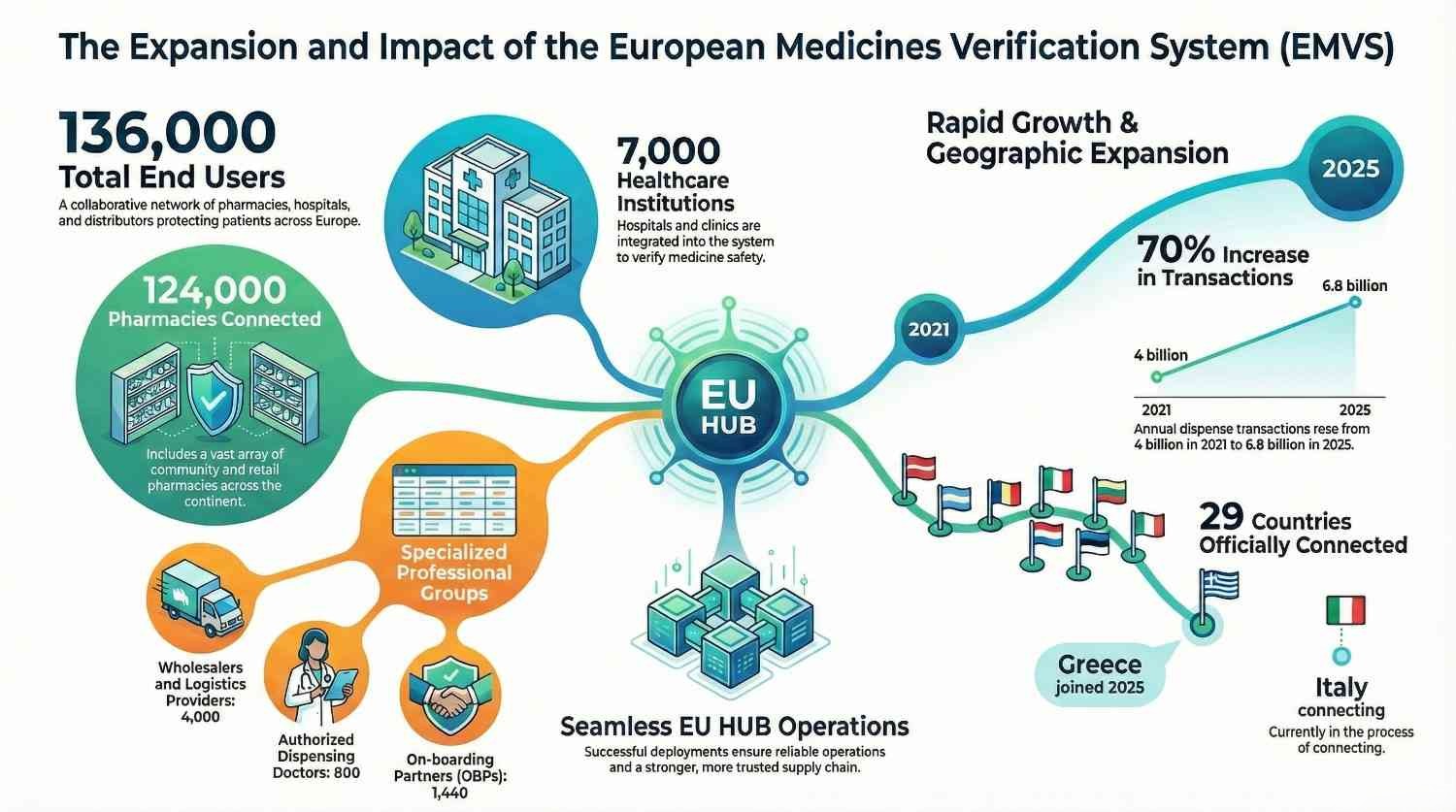
Celebrating 7 years of the European Medicines Verification System (EMVS)
On 9 February 2019, EMVS went live. On 9 February 2026,marks seven years of continuous operation securing the pharmaceu...
Price governance from Reactive to Proactive
Implementing Track and trace and through serialization allows regulatory bodies to transition from reactive pricing mode...
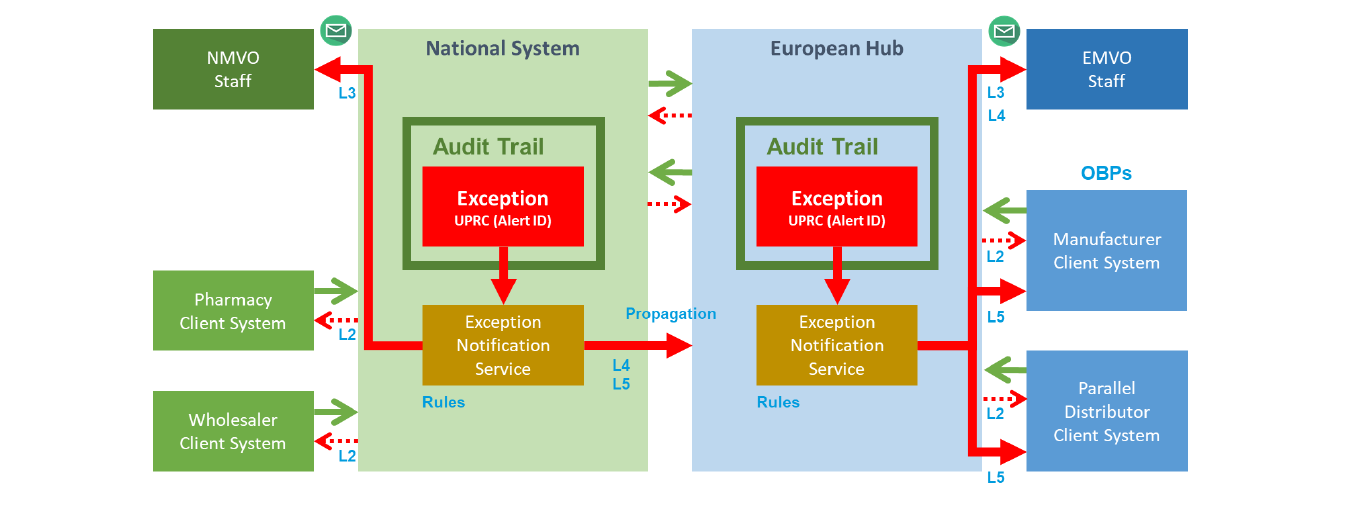
GS1 Data Matrix Exceptions
If you work across the Middle East and Africa, most serialization guidelines are straightforward.
Mobile Application in Track and Trace Reporting
Most Track & Trace hubs rely on traditional reporting channels: APIs for system-to-system integration, and sometime...
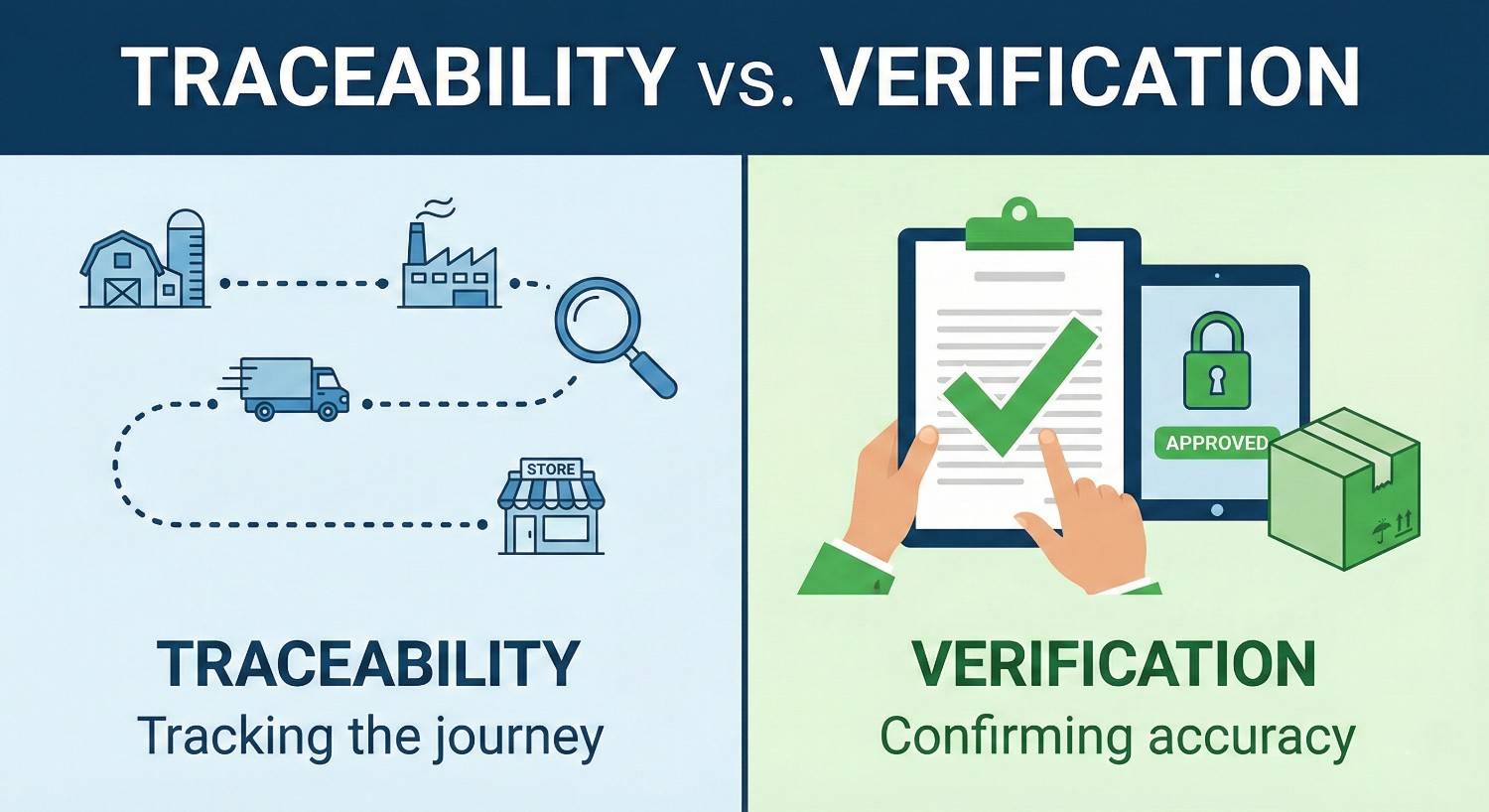
Traceability vs Verification
There is a common misconception around some key expressions related to Track & Trace, and this really needs clarificatio...
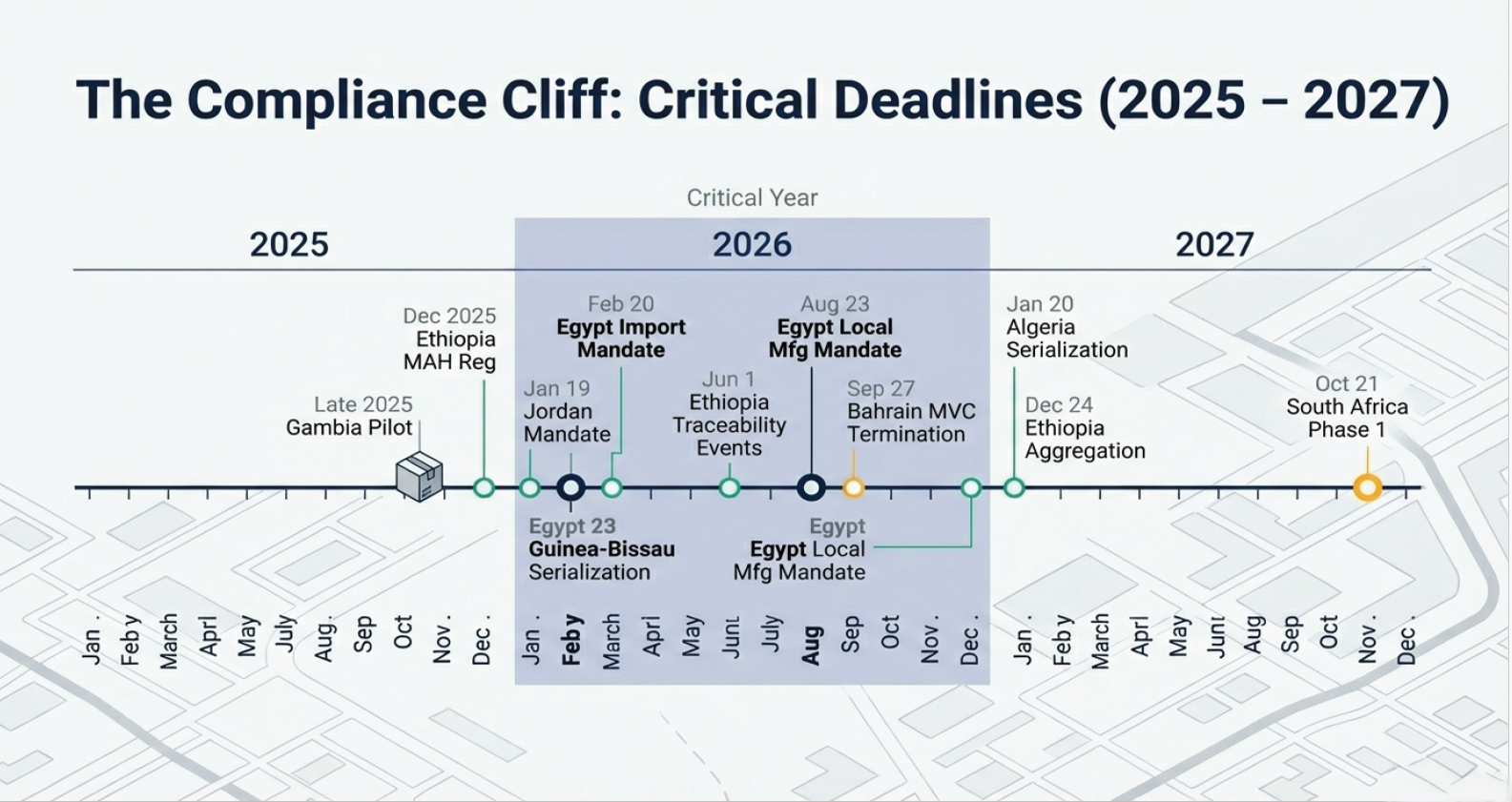
𝟐𝟎𝟐𝟔 𝐑𝐨𝐚𝐝𝐦𝐚𝐩 𝐟𝐨𝐫 𝐌𝐄𝐀 𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞
This year marks a historic surge in pharmaceutical traceability across Africa and the Middle East. For Marketing Authori...
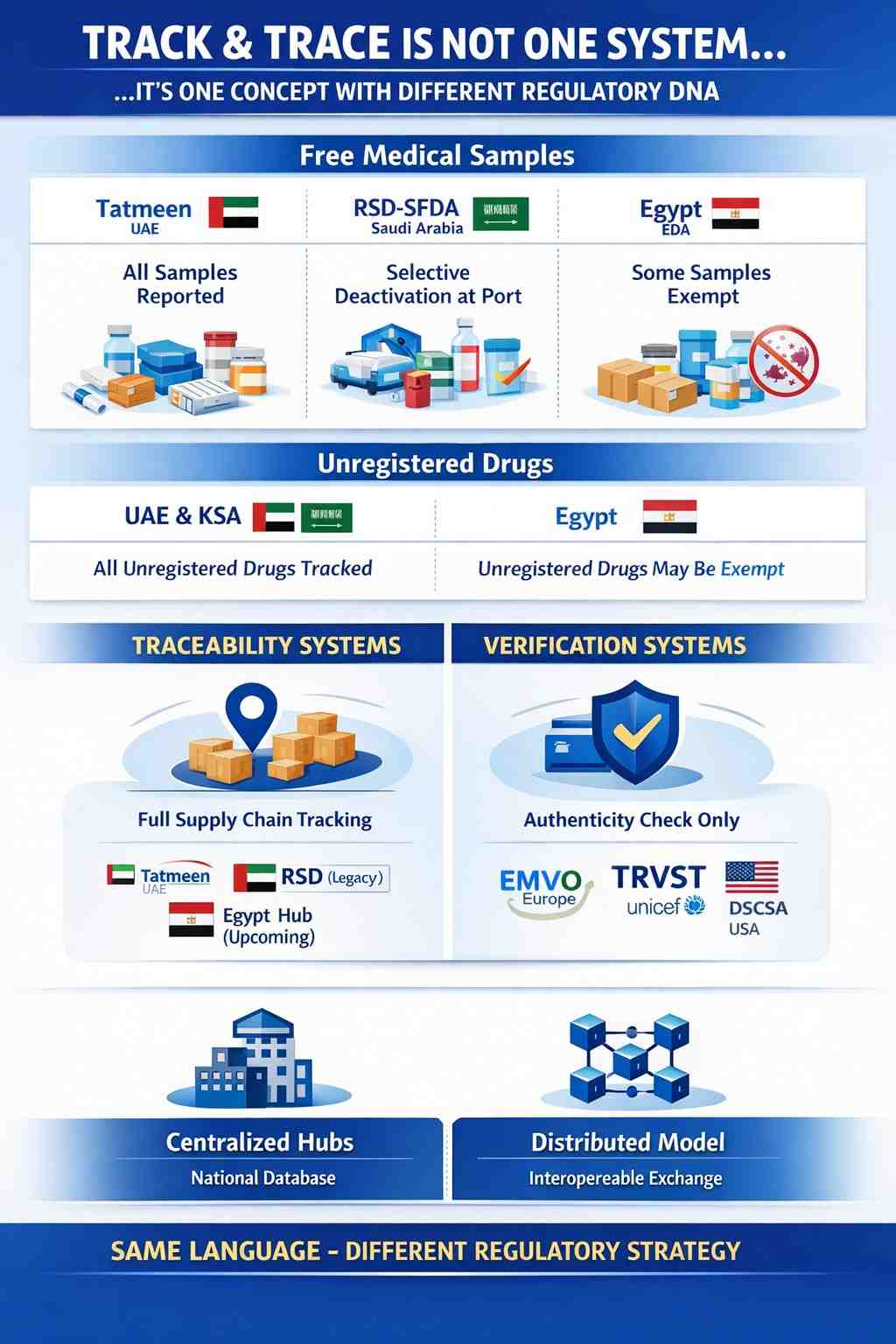
𝐔𝐧𝐩𝐨𝐩𝐮𝐥𝐚𝐫 𝐨𝐩𝐢𝐧𝐢𝐨𝐧: 𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 𝐢𝐬 𝐧𝐨𝐭 𝐨𝐧𝐞 𝐝𝐞𝐬𝐢𝐠𝐧𝐞𝐝 𝐬𝐲𝐬𝐭𝐞𝐦
One of the most common misunderstandings in pharmaceutical serialization is assuming that once you hear “Track & Trace”,...
𝐀𝐮𝐭𝐨𝐦𝐚𝐭𝐢𝐜 𝐈𝐝𝐞𝐧𝐭𝐢𝐟𝐢𝐜𝐚𝐭𝐢𝐨𝐧 & 𝐃𝐚𝐭𝐚 𝐂𝐚𝐩𝐭𝐮𝐫𝐞 (𝐀𝐈𝐃𝐂)
The silent engine behind every serious Track & Trace implementation.
Traceability Stories : Why Traceability-Patient Safety
The moment you see the surge of traceability announcements across the Middle East and Africa, one question likely spring...
𝐓𝐫𝐚𝐜𝐞𝐚𝐛𝐢𝐥𝐢𝐭𝐲 𝐒𝐭𝐨𝐫𝐢𝐞𝐬 : 𝐖𝐡𝐲 𝐓𝐫𝐚𝐜𝐞𝐚𝐛𝐢𝐥𝐢𝐭𝐲-Pricing Governance
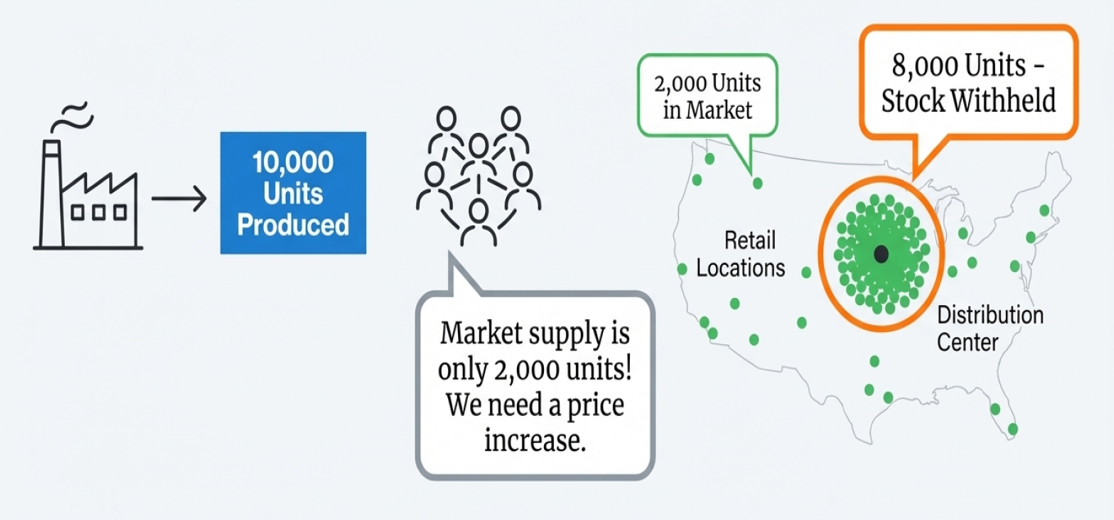
Pricing Pressure, Not Price Hikes: How Data Ends the Illusion of Drug Shortages
𝐖𝐇𝐎 𝐌𝐋𝐬,𝐆𝐁𝐓 𝐚𝐧𝐝 𝐀𝐟𝐫𝐢𝐜𝐚
World Health Organization considers medical products and health technologies one of the six core building blocks of heal...
Pharma Supply chain Gaps
How Counterfeit Medicines Slip Into the Supply Chain, And Why It Keeps Happening
How Traceability can serve to enhance Pharmacovigilance
Rethinking Point of view beyond compliance
The Hidden Cost of Choosing the Wrong Track & Trace Software
Selecting the wrong way to report the track & trace events can be one of the most expensive mistakes a company makes.
Is it time to rethink e-leaflets in alignment with pharmaceutical serialization?
Rethinking Point of view beyond compliance
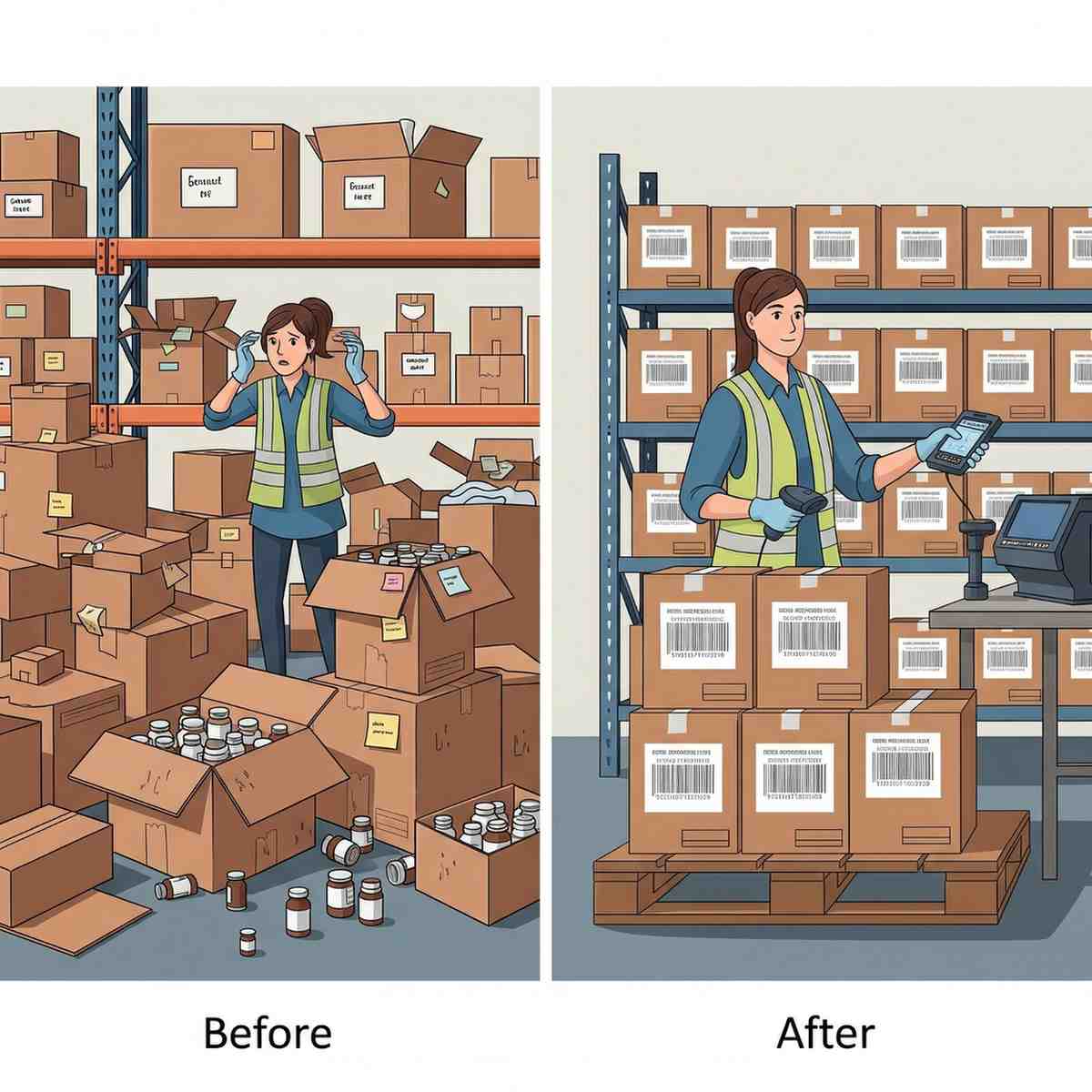
Serialization Impact on warehouse performance
Serialization is a game-changer for warehouse management. In this era where precision and traceability taking place in m...
𝐖𝐡𝐞𝐧 𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 𝐌𝐞𝐞𝐭𝐬 𝐋𝐞𝐠𝐚𝐜𝐲 𝐒𝐲𝐬𝐭𝐞𝐦𝐬 (𝐌𝐨𝐦𝐞𝐧𝐭 𝐎𝐟 𝐓𝐫𝐮𝐭𝐡)
As countries across the region push forward with national serialization mandates (like Tatmeen in UAE, RSD in KSA, and N...
The Weak Links in the Global Pharma Supply Chain
Insights from WHO, Pharmaceutical Security Institute (PSI) Situation Reports.
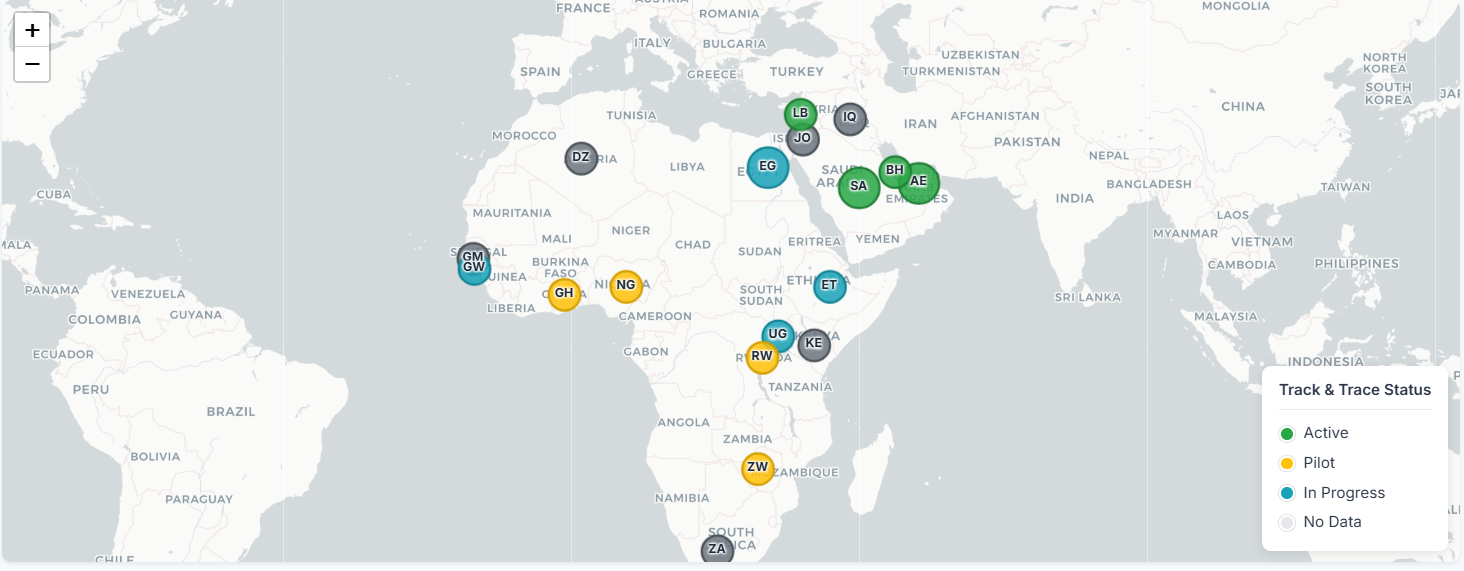
Pharmaceutical Track & Trace Momentum Across Middle East & Africa
In just the last two quarters of 2025, nearly 10 countries across the Middle East and Africa have announced launching th...
Aggregation as a Service (AaaS)
Pharma manufacturers today face a tough decision when it comes to track & trace compliance
Track vs Trace in pharmaceuticals
Track: Monitoring the current location of products in real-time. Tr...
When Your Current WMS / HIS / POS / ERP Meets Track & Trace: Adapt or Struggle
The Track & Trace journey has reached a major milestone , it’s already live in KSA, UAE, and Bahrain, with rollouts star...
Country Specific (27 articles)
FDA’s 12-Digit NDC Final Rule
The FDA has officially set the clock. On March 5, 2026, the Final Rule standardizing the National Drug Code (NDC) to a u...
EMVO Systems Updates | May 2026
Explore the latest improvements across the European Medicines Verification System (EMVS), strengthening performance, usa...
𝐔𝐀𝐄’𝐬 𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 (𝐓𝐚𝐭𝐦𝐞𝐞𝐧) 𝐎𝐯𝐞𝐫𝐯𝐢𝐞𝐰
In December 2022, the UAE activated one of the region’s most structured pharmaceutical traceability ecosystems:#Tatmeen.
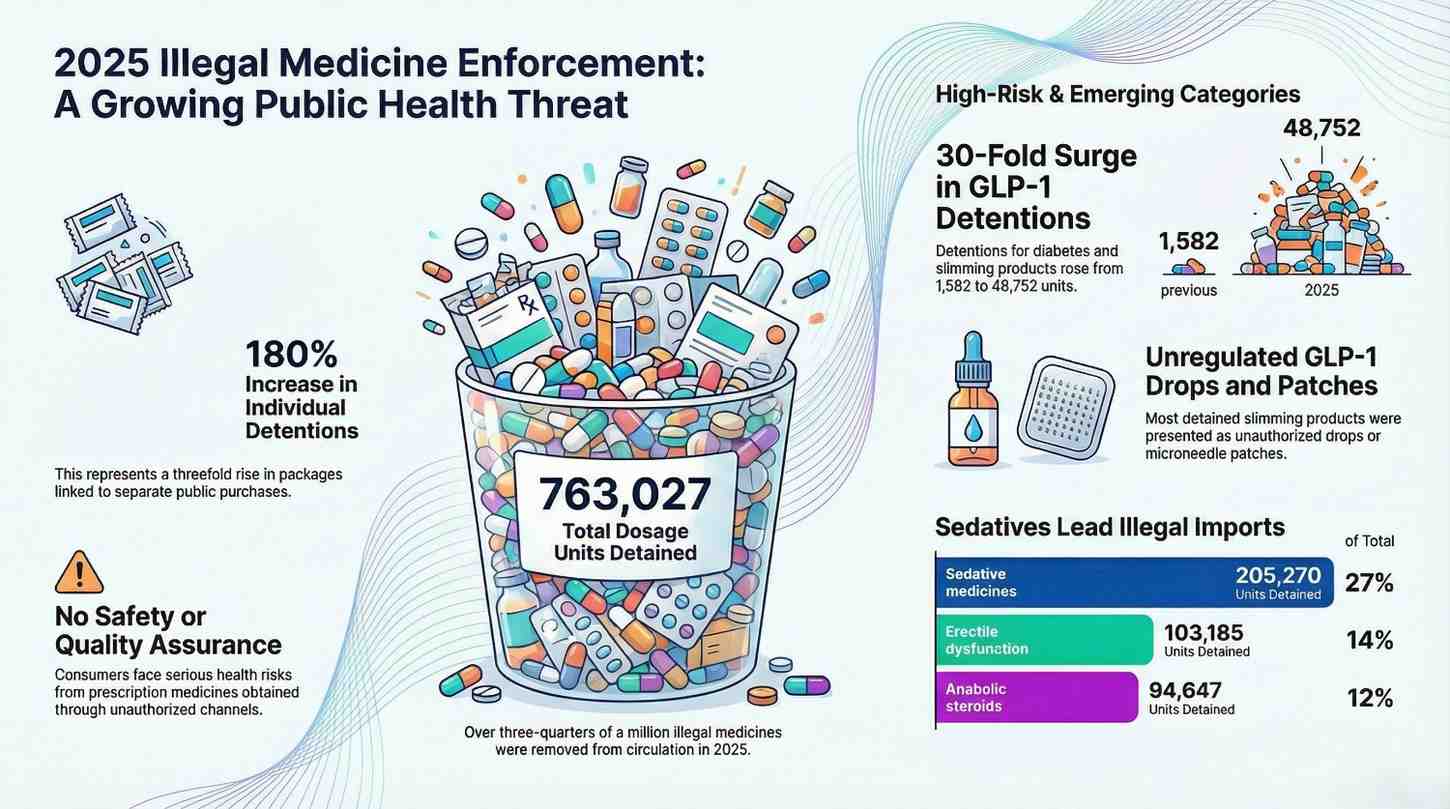
Over 750,000 illegal medicine units detained in Ireland at 2025
Health Products Regulatory Authority (HPRA) Ireland regulatory authority announced Over 750,000 illegal medicine units d...
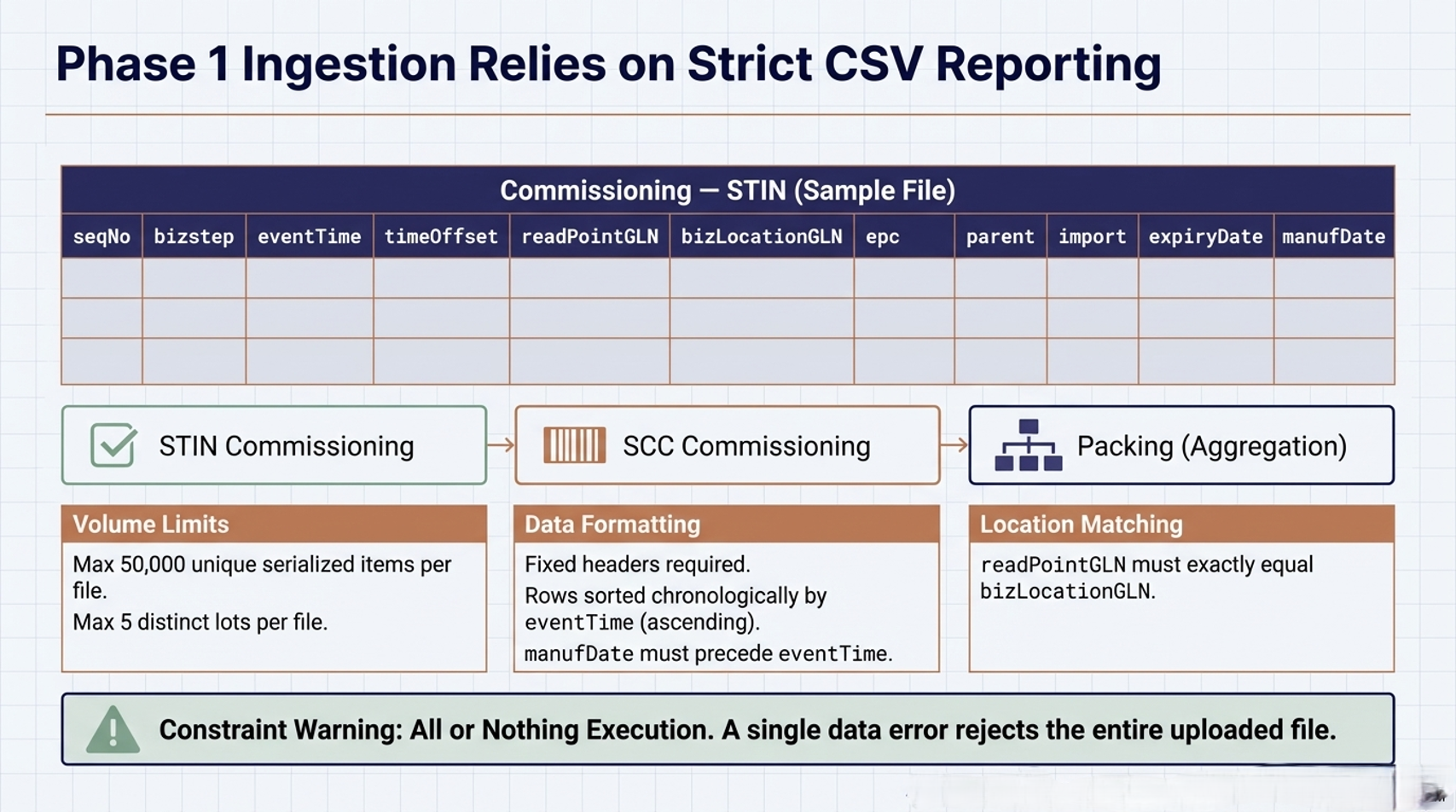
Turning the CSV Template into a Shipment Story in EPTTS
Egypt Pharma Track & Trace (Phase 1),EPTTS required CSV file reporting in this phase
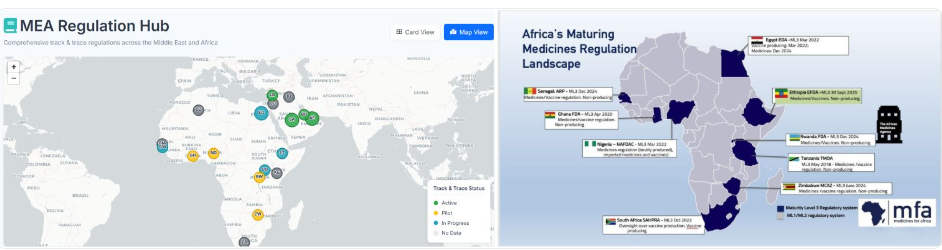
Why Traceability mandates linked to WHO Maturity levels
Across Africa, something important is happening in regulatory systems,and it’s not getting enough attention.
WHO Maturity Levels and Traceability
Across the MEA region, national regulatory authorities (NRAs) are increasingly accelerating the adoption of pharmaceutic...
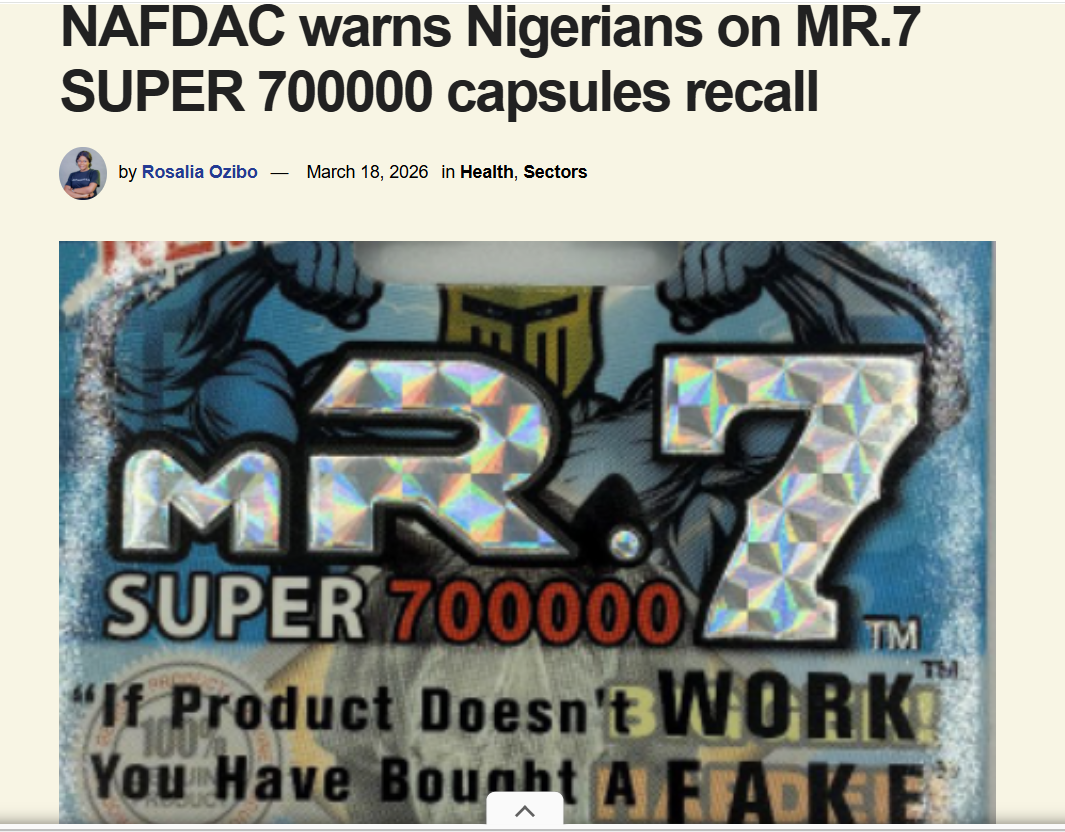
𝐑𝐞𝐜𝐚𝐥𝐥 𝐒𝐧𝐚𝐩𝐬𝐡𝐨𝐭 𝐢𝐧 𝐀𝐟𝐫𝐢𝐜𝐚
In just less than a month , multiple pharmaceutical products were recalled or flagged across several countries in #Afric...
Behind the Beep: EMVS Digital Guard
You’re standing at a pharmacy. A simple beep… and the pack is handed to you. Seems routine. It’s not.
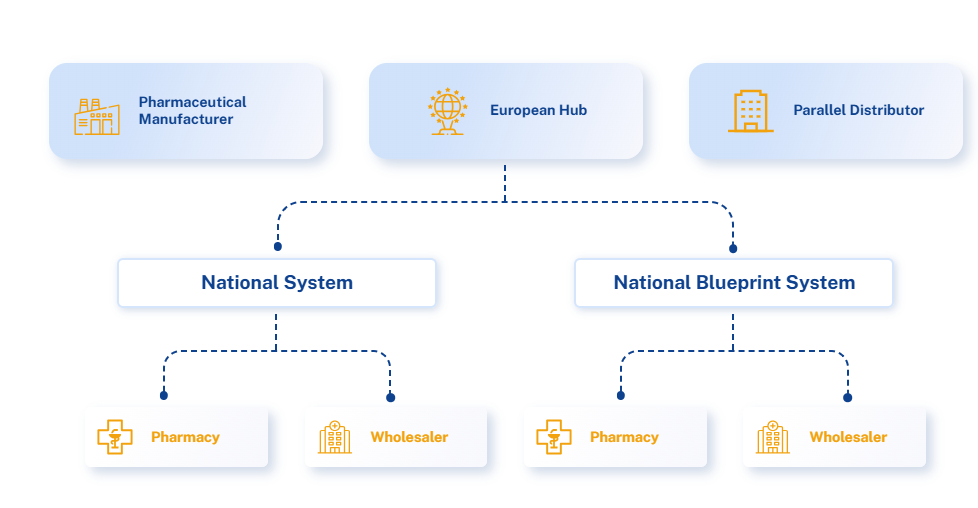
𝐔𝐧𝐝𝐞𝐫𝐬𝐭𝐚𝐧𝐝𝐢𝐧𝐠 𝐭𝐡𝐞 𝐄𝐌𝐕𝐎 𝐌𝐨𝐝𝐞𝐥 ,𝐓𝐡𝐞 𝐌𝐨𝐬𝐭 𝐒𝐭𝐫𝐮𝐜𝐭𝐮𝐫𝐞𝐝 𝐄𝐱𝐚𝐦𝐩𝐥𝐞 𝐨𝐟 𝐚 𝐕𝐞𝐫𝐢𝐟𝐢𝐜𝐚𝐭𝐢𝐨𝐧 𝐒𝐲𝐬𝐭𝐞𝐦 𝐢𝐧 𝐏𝐡𝐚𝐫𝐦𝐚
Let’s start with a critical clarification: 𝑵𝒐𝒕 𝒂𝒍𝒍 𝒔𝒚𝒔𝒕𝒆𝒎𝒔 𝒂𝒓𝒆 𝒕𝒉𝒆 𝒔𝒂𝒎𝒆.
𝐅𝐃𝐀 𝐖𝐚𝐫𝐧𝐢𝐧𝐠 𝐋𝐞𝐭𝐭𝐞𝐫: 𝐖𝐡𝐞𝐧 𝐂𝐨𝐦𝐩𝐥𝐢𝐚𝐧𝐜𝐞 𝐅𝐚𝐢𝐥𝐬
A recent warning letter issued by the U.S. Food and Drug Administration FDA to a dispenser
𝐀𝐟𝐫𝐢𝐜𝐚 𝐈𝐬 𝐖𝐡𝐞𝐫𝐞 𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 𝐆𝐞𝐭𝐬 𝐑𝐞𝐚𝐥
The Track & Trace conversation is shifting… and right now, the real momentum is happening in Africa.
Italy started their journey to join the flagship verification system EMVO
Italy, which already had a unique identification system for medicinal products, was granted an extension until 2025
Some Notes about march guidelines announced by EDA
Some additional notes on the guidelines announced at the beginning of March regarding the drug tracking system in Egypt ...
March Status of Pharmaceutical Track & Trace in Egypt
Egypt’s pharmaceutical traceability journey is now moving from planning to real execution.
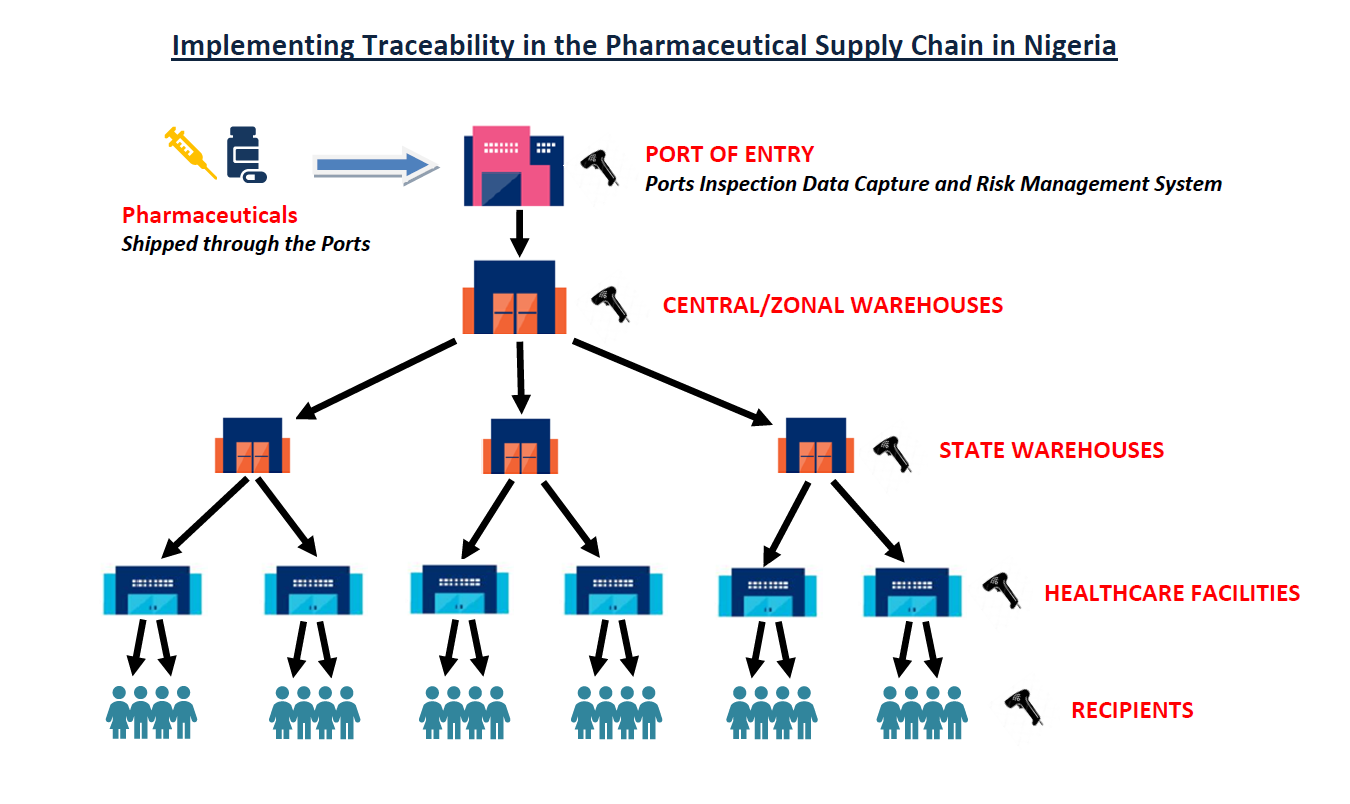
𝐏𝐡𝐚𝐫𝐦𝐚𝐜𝐞𝐮𝐭𝐢𝐜𝐚𝐥 𝐓𝐫𝐚𝐜𝐞𝐚𝐛𝐢𝐥𝐢𝐭𝐲 𝐢𝐧 𝐍𝐢𝐠𝐞𝐫𝐢𝐚 – 𝐒𝐭𝐚𝐭𝐮𝐬 𝐔𝐩𝐝𝐚𝐭𝐞
The regulatory backbone is the National Agency for Food and Drug Administration and Control (NAFDAC) its main objectiv...
𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 𝐌𝐨𝐦𝐞𝐧𝐭𝐮𝐦 𝐢𝐧 𝐀𝐟𝐫𝐜𝐢𝐚
It is fair to state that Track & Trace in Africa has entered a true acceleration phase.
𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 𝐢𝐧 𝐒𝐚𝐮𝐝𝐢 𝐀𝐫𝐚𝐛𝐢𝐚( 𝐅𝐫𝐨𝐦 𝐓𝐫𝐚𝐜𝐞𝐚𝐛𝐢𝐥𝐢𝐭𝐲 𝐭𝐨 𝐕𝐞𝐫𝐢𝐟𝐢𝐜𝐚𝐭𝐢𝐨𝐧)
Let’s shed light on how pharmaceutical traceability evolved in Saudi Arabia under the national platform SFDA - هيئة الغذ...
𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 𝐈𝐦𝐩𝐥𝐞𝐦𝐞𝐧𝐭𝐚𝐭𝐢𝐨𝐧 𝐑𝐨𝐚𝐝𝐦𝐚𝐩 𝐢𝐧 𝐊𝐞𝐧𝐲𝐚
The standard announced in last septmber covers all medicinal, cosmetic, and personal care products consumed, injected, o...
Pre Track & Trace Implementation in Egypt, what you need to do?
With the official start of pharmaceutical track & trace in Egypt scheduled for February for imported medicines, and a tr...
EDA Track & Trace Q&A – Detailed Clarification
these were some Q&A published before in October 2025 by EDA
Warehouses Must Operationally Prepare for SSCC-Based Handling Under EDA’s Decision 804
Under Decision 804, logistics movements between manufacturers, distributors and pharmacies are expected to be reported a...
Key Market Insights and Technical Reflections on Egypt’s Track & Trace Draft Guidelines
Reading from EDA announced draft guidelines adn some thought about the Egyptian pharmaceutical market architecture
Substandard and falsified medical products in south Africa
Highlighting some points in the article shared by head of regulatory compliance in SAHPRA
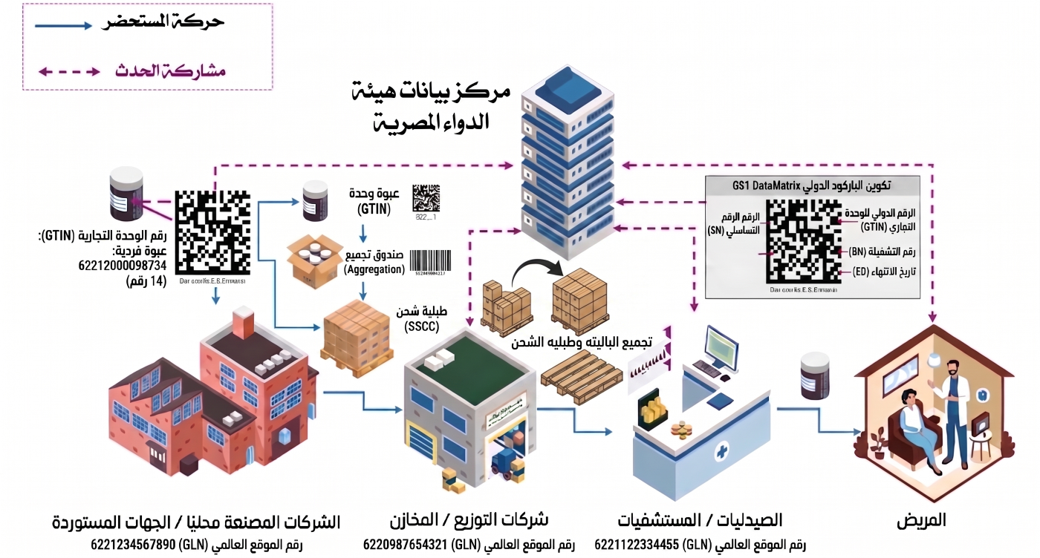
Understanding Egypt’s Pharma Supply Chain
Earlier this month, the Egyptian Drug Authority (EDA) officially announced in the official gazette the establishment of ...
Uzbekistan’s National Hub new requirement
Update in serial generation of items imported to Uzbekistan
EPCIS Basics (7 articles)
Introduction to EPCIS
A comprehensive introduction to Electronic Product Code Information Services (EPCIS) and its role in pharmaceutical trac...
EPCIS Event Types Explained
Deep dive into the four EPCIS event types: ObjectEvent, AggregationEvent, TransactionEvent, and TransformationEvent.
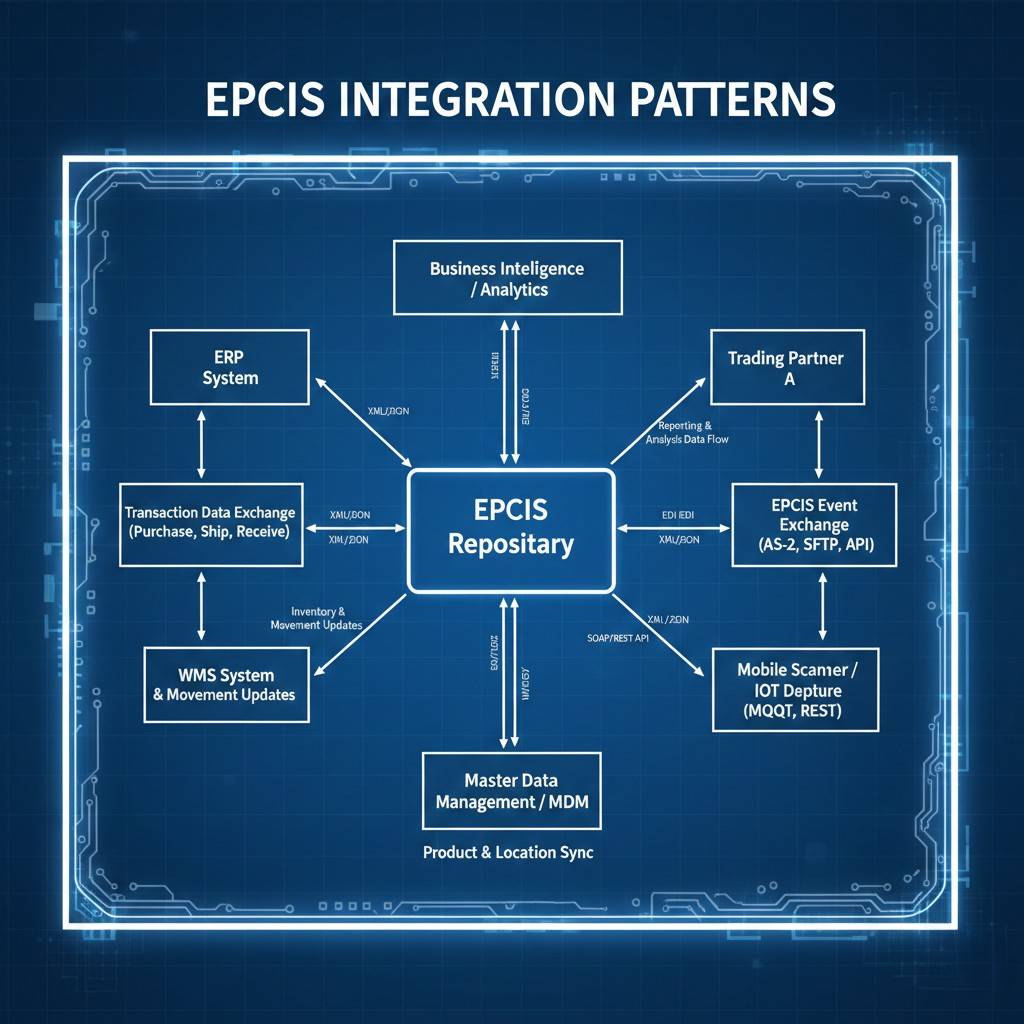
EPCIS Integration Patterns
Technical guide to integrating EPCIS systems: API patterns, file-based exchange, and real-time vs batch processing.
EPCIS Business Steps and Dispositions
Understanding the business context in EPCIS: business steps, dispositions, and how they describe the 'why' of events.
EPCIS Data Formats: XML vs JSON
Comparing EPCIS XML and JSON formats, structure differences, and when to use each format.
Common EPCIS Errors and Solutions
Troubleshooting guide for common EPCIS implementation errors with practical solutions.
EPCIS Core Business Vocabularies (CBV)
Understanding GS1 Core Business Vocabularies and how they provide standardized terminology for EPCIS events.
GS1 Standards (6 articles)
Understanding GS1 Identifiers
Complete guide to GS1 identification keys: GTIN, SSCC, SGTIN, GLN and their role in pharmaceutical serialization.
Impact of GS1 data matrix on operational enhancements
Rethinking in the value of GS1 data matrix and how it can impact the entire operation
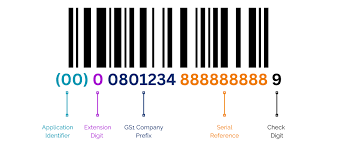
GS1 DataMatrix Barcodes for Pharmaceuticals
Learn about GS1 DataMatrix 2D barcodes, Application Identifiers, and encoding requirements for pharmaceutical packaging.
GCP The Hidden Backbone Behind GTIN, GLN, and SSCC
Article shows the role of GCP in other company components
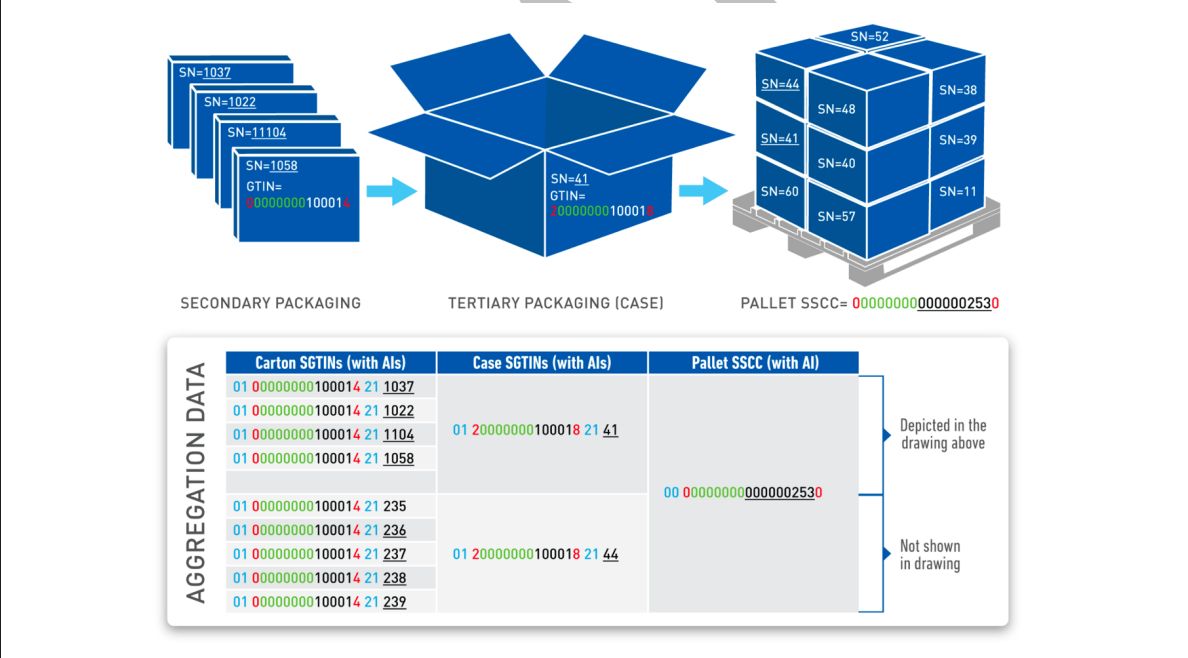
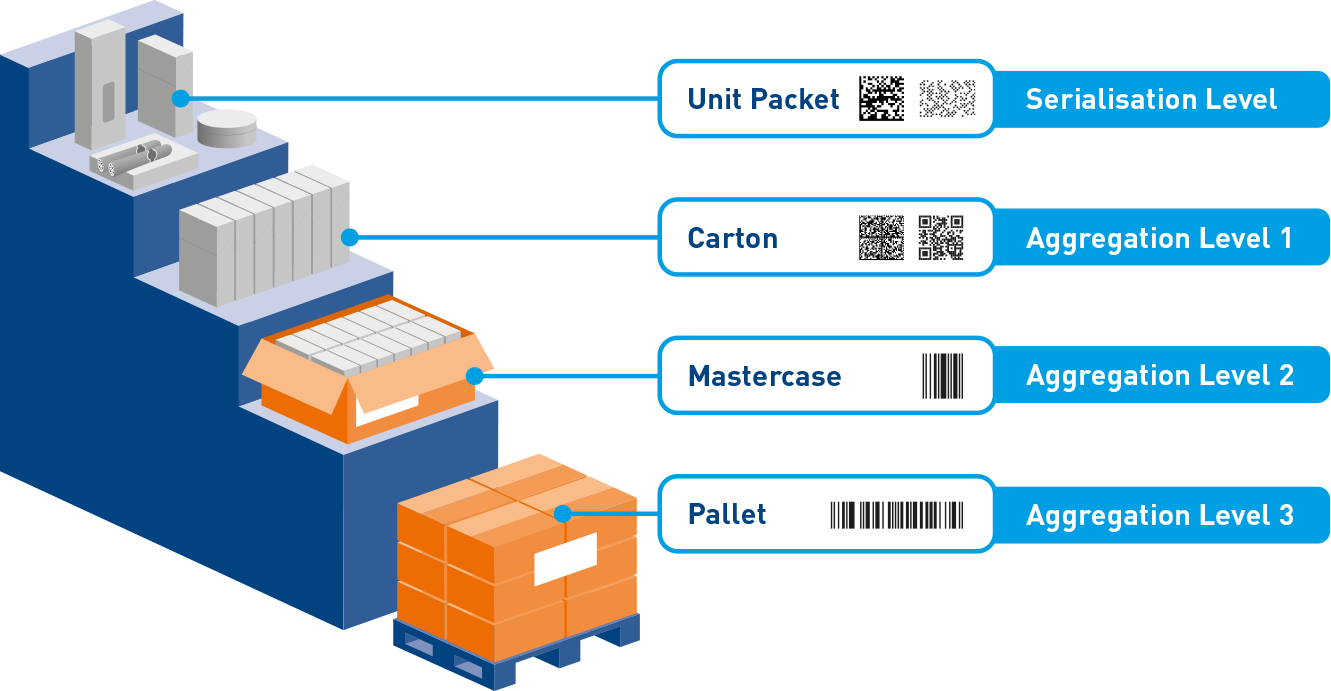
Pharmaceutical Serialization Levels
Understanding unit, case, and pallet level serialization requirements and aggregation hierarchies.
GS1 Company Prefix: Getting Started
How to obtain a GS1 Company Prefix and use it to generate valid GTINs, SSCCs, and other identifiers.
Implementation Guides (8 articles)
Master Data is the #1 Hidden Risk in Track & Trace
(GLNs, GTINs… and the problems no one plans for)
Why Master Data Is the Real Starting Point of Track & Trace
When we took a look on EDA announcements of track and trace and focus on Wholesalers segment ,you need to rethink to ...
National Hub Compliance Starting Point
one of the biggest operational challenges in Track & Trace , how to start compliance reporting when there is already exi...
Change management in Track and Trace implementation
Track & Trace is not only to apply technology, but it is a mindset.
In choosing a Track & Trace Solution, Do You Really Know What You Need?
With national serialization and reporting regulations rolling out across the region , every company faces the same chall...
Different hubs… one T&T core three different sample policies
Pharmaceutical track & trace is one global language…but every market speaks it with a different accent.
Aggregation Hierarchy Best Practices
Best practices for building and maintaining aggregation relationships from items to cases to pallets.
Testing Your EPCIS Implementation
Comprehensive testing guide for EPCIS implementations including validation tools, test scenarios, and certification.
Operational Challenges (7 articles)
Acting as MAH & CMO challenges
I’m seeing more and more manufacturers stepping into a dual identity: you’re a Marketing Authorization Holder (MAH) for...
Post-Implementation of track and trace (Operational Reality)
When analyzing manufacturers and distributors who have already implemented pharmaceutical track & trace, one reality sta...
Cost of change after Track & Trace implementation
Changing your serialization or aggregation machine vendor, or changing your track and trace software provider, or migrat...
Forward-Looking T&T applying model (silent operational challenges)
In many markets, the adoption of Forward-Looking Traceability which means ( determine deadline for applying track and t...
New Traceability Challenge (One Shipment, Double Reporting)
Article discuss the new operational challenge that appear after momentum of traceability announcements regionally
Distributors and wholesalers T&T implementation challenges
An eye on the T&T implementation challenges in Distributors and wholesalers segment
Impact of Serialization on Order Fulfillment Accuracy
Article about impact of serialization on Order Fulfillment Accuracy