Regulatory News
Latest updates from regulatory authorities worldwide
SFDA penalized 19 pharmaceutical establishments
Saudi Food and Drug Authority has penalized 19 pharmaceutical establishments with fines exceeding SAR 5 million during A...
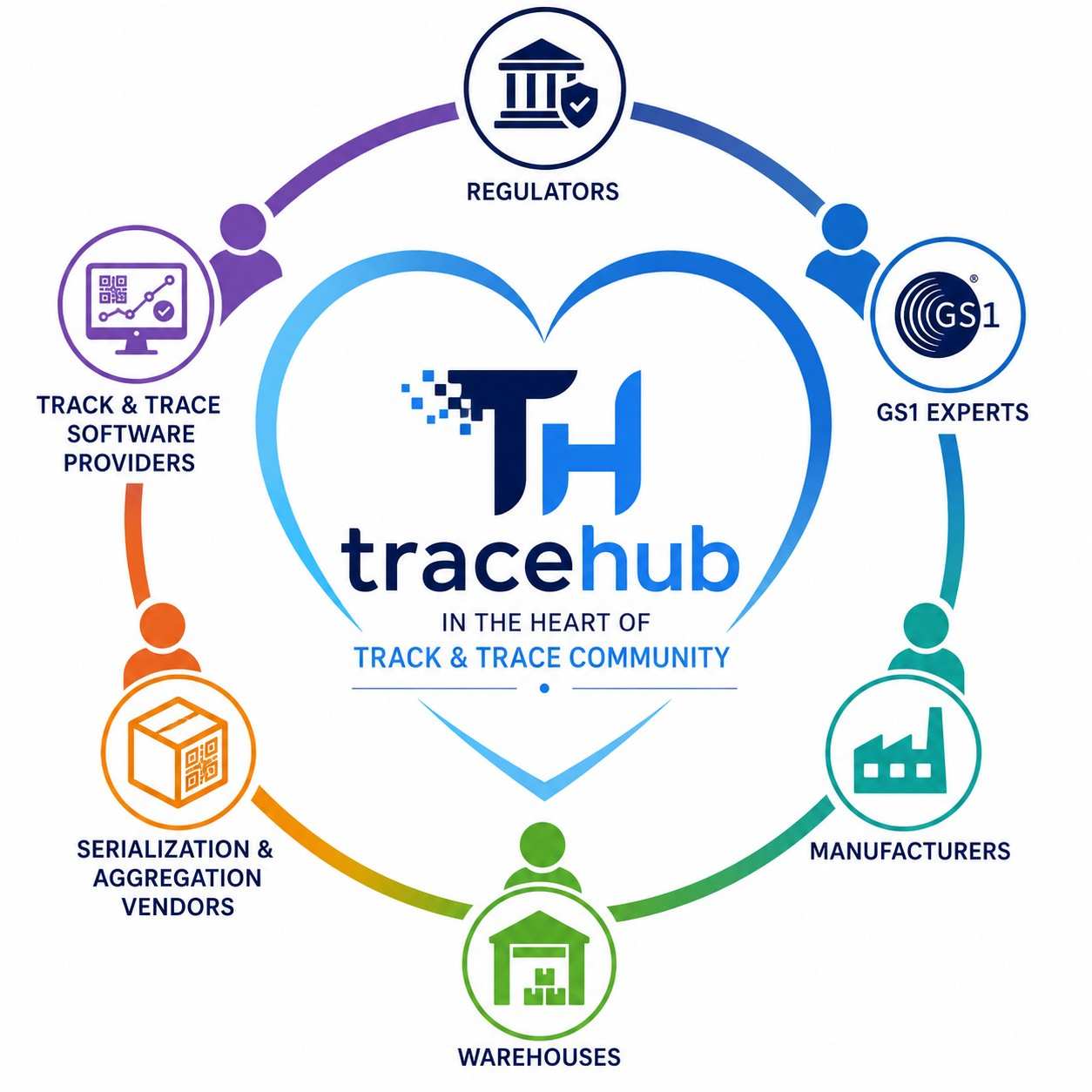
TraceHub in the heart of track and trace community
When I first started my journey in pharmaceutical Track & Trace, I faced two major challenges. The first challenge w...
Track & Trace in Libya
Libya is taking a significant new step toward strengthening pharmaceutical governance and supply chain transparency. The...
TraceHub Global Virtual Community Meeting Agenda
I’m thrilled to invite you to join the 1st Ever Global Track & Trace Community Event, a unique virtual gathering bri...
Launch of TATABU Medical Devices & Supplies Traceability System
The National Health Regulatory Authority (NHRA) in the Kingdom of Bahrain has officially announced the launch of the TAT...
Calling for Track & Trace Initiative | Global Virtual Community Meeting.
I’ve been thinking lately... with the growing global momentum around pharmaceutical Track & Trace, maybe this is the...
A New Digital System to Monitor the Pharmaceutical Market in Algeria
A New Digital System to Monitor the Pharmaceutical Market in Algeria In a new step to strengthen health security and ens...
EDA release updated version from EPTTS technical FAQs phase 1..V3
The main changes in point 5 and 7 5-Data Matrix Encoding Requirements (changes in exp date to align with GS1 standards) ...
Ahmed Dawood wrote .. "Why TraceHub?"
Once I started my journey in the pharmaceutical track & trace field, one challenge stood out more than anything else...
Reminder: Bahrain Traceability Hub Transition
On October last year, circular issued by NHRA confirms a critical update for all pharmaceutical supply chain stakeholder...
Track & Trace Updates in Egypt (April Edition)
Good morning, let’s take a quick look at the latest updates on pharmaceutical track & trace in Egypt so far... and a...
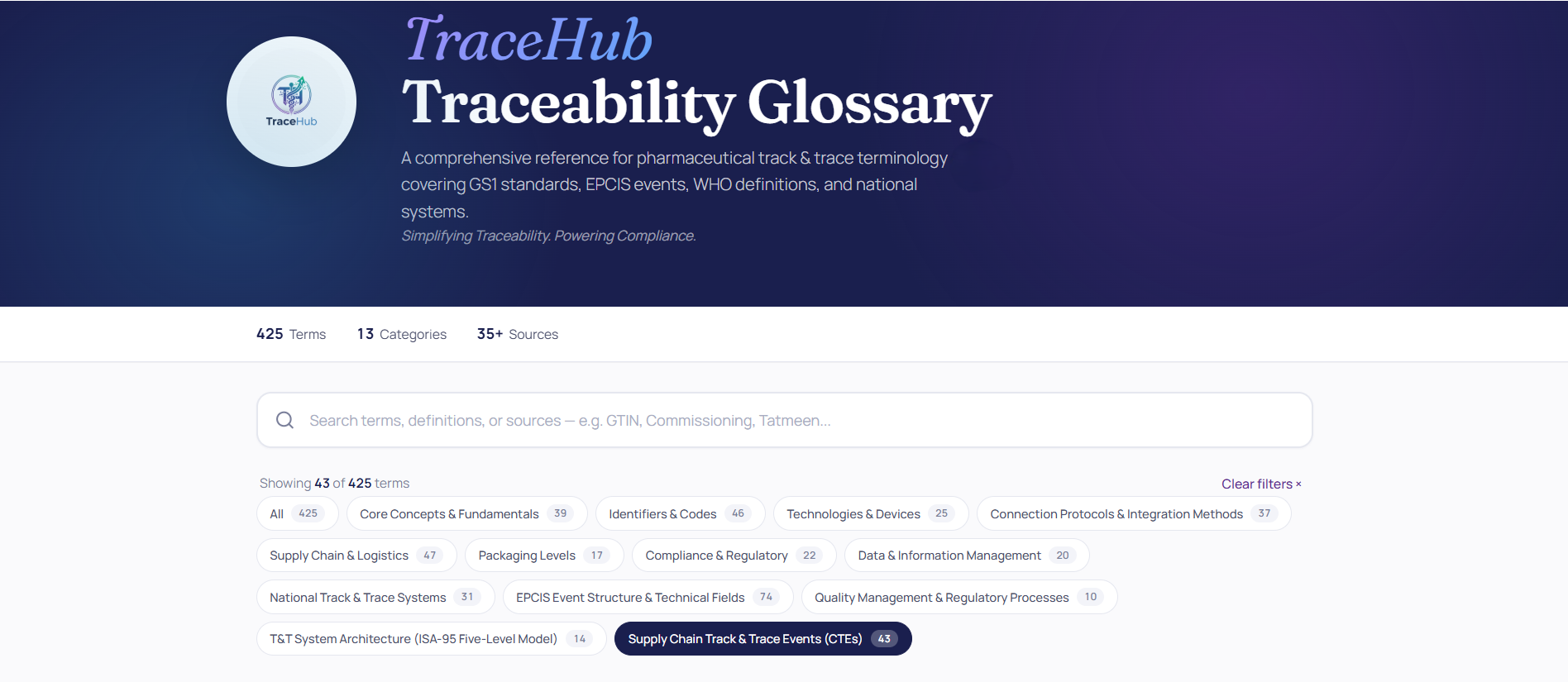
TraceHub Traceability Glossary... Now Live
We’re proud to announce that the first release of the TraceHub Traceability Glossary is now officially live. 📌 13 speci...