Knowledge Base for serialization engineers.
Learn about EPCIS, GS1 Standards, and Track & Trace implementation
Italy started their journey to join the flagship verification system EMVO
Italy, which already had a unique identification system for medicinal products, was granted an extension until 2025
EMVO Systems Updates | May 2026
Explore the latest improvements across the European Medicines Verification System (EMVS), strengthening performance, usa...
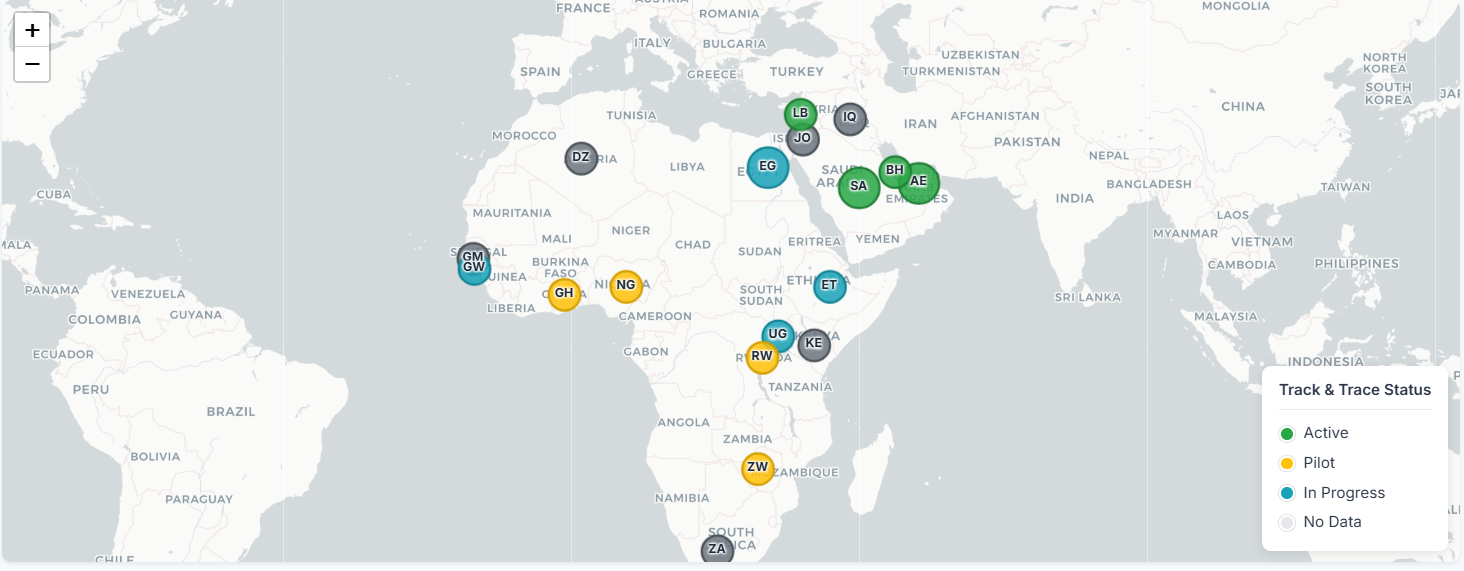
𝐀𝐟𝐫𝐢𝐜𝐚 𝐈𝐬 𝐖𝐡𝐞𝐫𝐞 𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 𝐆𝐞𝐭𝐬 𝐑𝐞𝐚𝐥
The Track & Trace conversation is shifting… and right now, the real momentum is happening in Africa.
𝐅𝐃𝐀 𝐖𝐚𝐫𝐧𝐢𝐧𝐠 𝐋𝐞𝐭𝐭𝐞𝐫: 𝐖𝐡𝐞𝐧 𝐂𝐨𝐦𝐩𝐥𝐢𝐚𝐧𝐜𝐞 𝐅𝐚𝐢𝐥𝐬
A recent warning letter issued by the U.S. Food and Drug Administration FDA to a dispenser
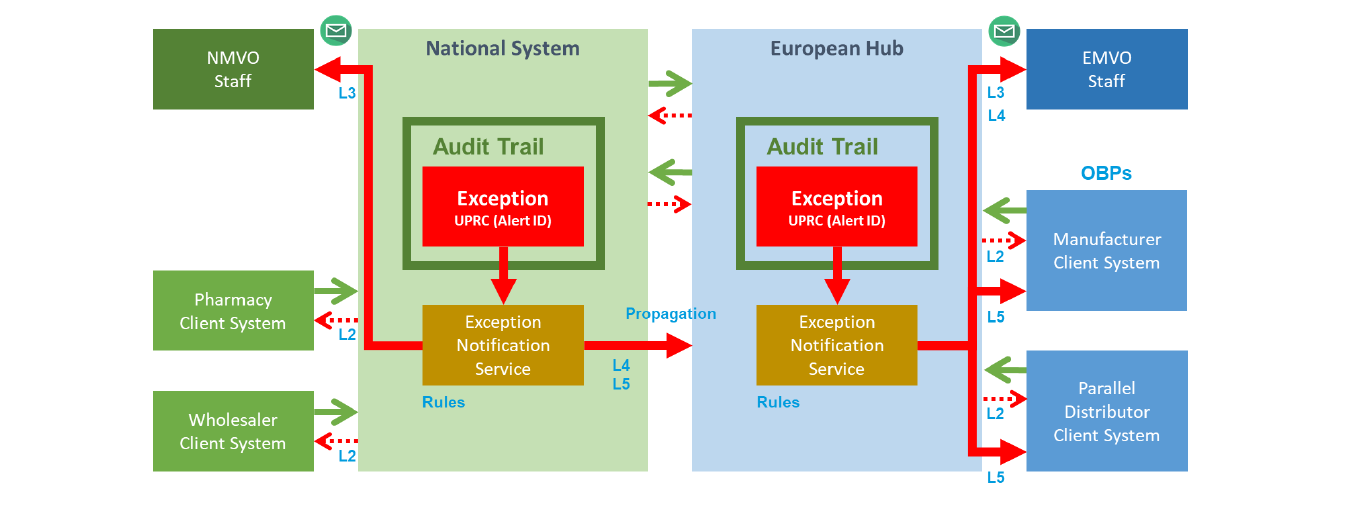
𝐔𝐧𝐝𝐞𝐫𝐬𝐭𝐚𝐧𝐝𝐢𝐧𝐠 𝐭𝐡𝐞 𝐄𝐌𝐕𝐎 𝐌𝐨𝐝𝐞𝐥 ,𝐓𝐡𝐞 𝐌𝐨𝐬𝐭 𝐒𝐭𝐫𝐮𝐜𝐭𝐮𝐫𝐞𝐝 𝐄𝐱𝐚𝐦𝐩𝐥𝐞 𝐨𝐟 𝐚 𝐕𝐞𝐫𝐢𝐟𝐢𝐜𝐚𝐭𝐢𝐨𝐧 𝐒𝐲𝐬𝐭𝐞𝐦 𝐢𝐧 𝐏𝐡𝐚𝐫𝐦𝐚
Let’s start with a critical clarification: 𝑵𝒐𝒕 𝒂𝒍𝒍 𝒔𝒚𝒔𝒕𝒆𝒎𝒔 𝒂𝒓𝒆 𝒕𝒉𝒆 𝒔𝒂𝒎𝒆.
Behind the Beep: EMVS Digital Guard
You’re standing at a pharmacy. A simple beep… and the pack is handed to you. Seems routine. It’s not.
𝐑𝐞𝐜𝐚𝐥𝐥 𝐒𝐧𝐚𝐩𝐬𝐡𝐨𝐭 𝐢𝐧 𝐀𝐟𝐫𝐢𝐜𝐚
In just less than a month , multiple pharmaceutical products were recalled or flagged across several countries in #Afric...
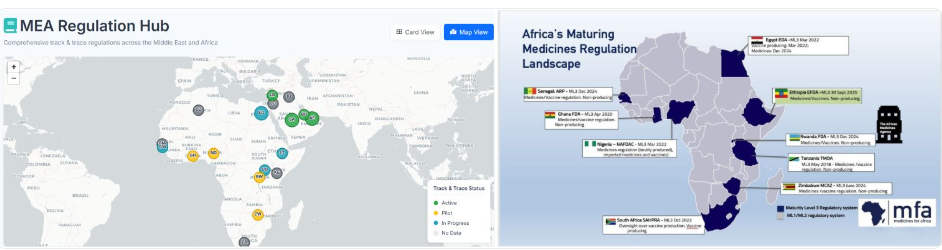
WHO Maturity Levels and Traceability
Across the MEA region, national regulatory authorities (NRAs) are increasingly accelerating the adoption of pharmaceutic...
Why Traceability mandates linked to WHO Maturity levels
Across Africa, something important is happening in regulatory systems,and it’s not getting enough attention.
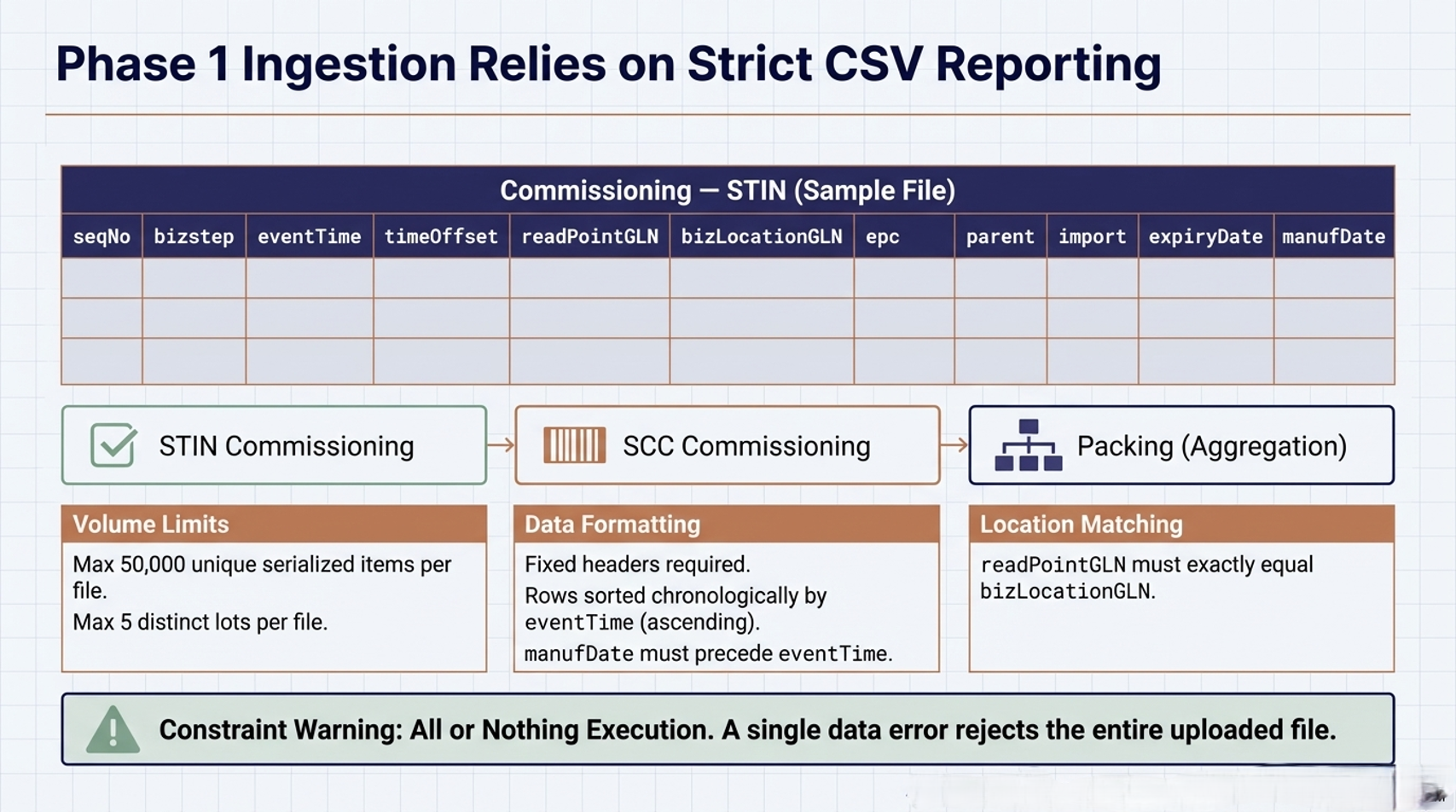
Turning the CSV Template into a Shipment Story in EPTTS
Egypt Pharma Track & Trace (Phase 1),EPTTS required CSV file reporting in this phase
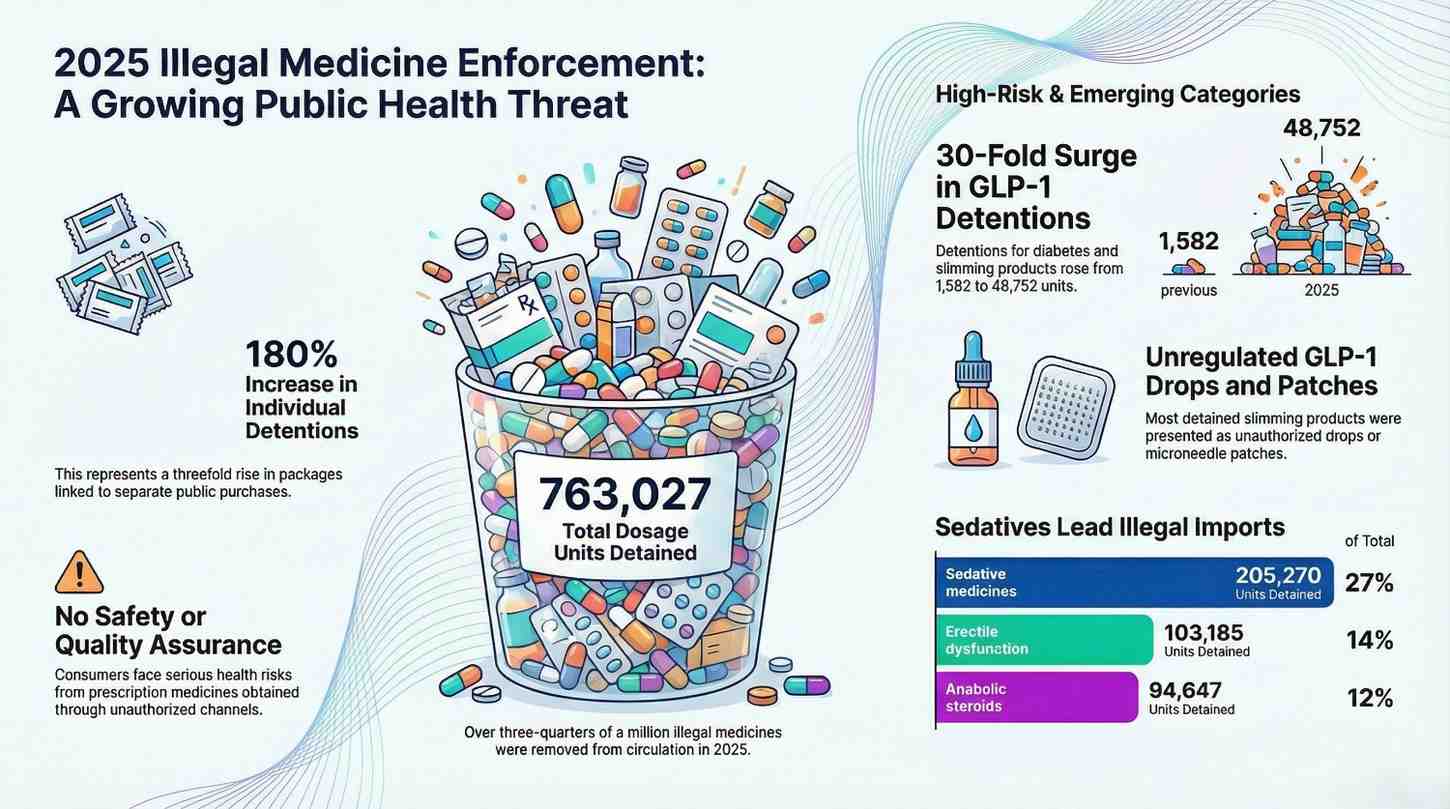
Over 750,000 illegal medicine units detained in Ireland at 2025
Health Products Regulatory Authority (HPRA) Ireland regulatory authority announced Over 750,000 illegal medicine units d...
𝐔𝐀𝐄’𝐬 𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 (𝐓𝐚𝐭𝐦𝐞𝐞𝐧) 𝐎𝐯𝐞𝐫𝐯𝐢𝐞𝐰
In December 2022, the UAE activated one of the region’s most structured pharmaceutical traceability ecosystems:#Tatmeen.
FDA’s 12-Digit NDC Final Rule
The FDA has officially set the clock. On March 5, 2026, the Final Rule standardizing the National Drug Code (NDC) to a u...
Some Notes about march guidelines announced by EDA
Some additional notes on the guidelines announced at the beginning of March regarding the drug tracking system in Egypt ...
March Status of Pharmaceutical Track & Trace in Egypt
Egypt’s pharmaceutical traceability journey is now moving from planning to real execution.
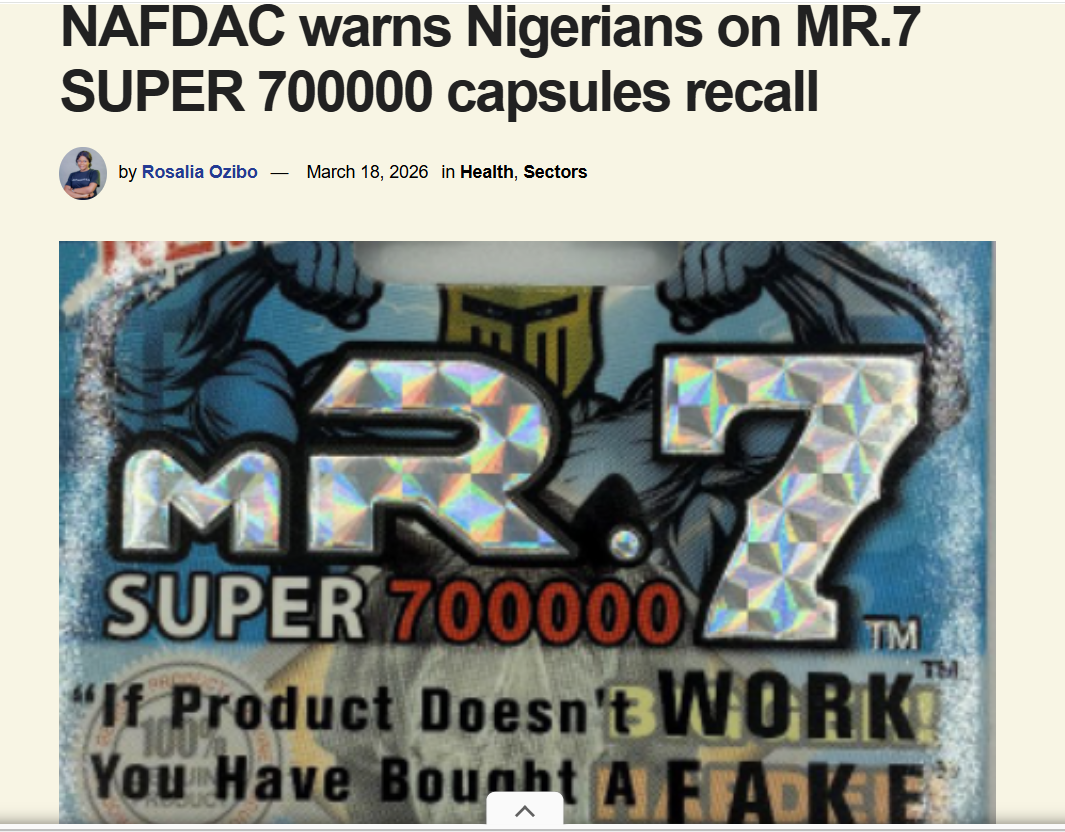
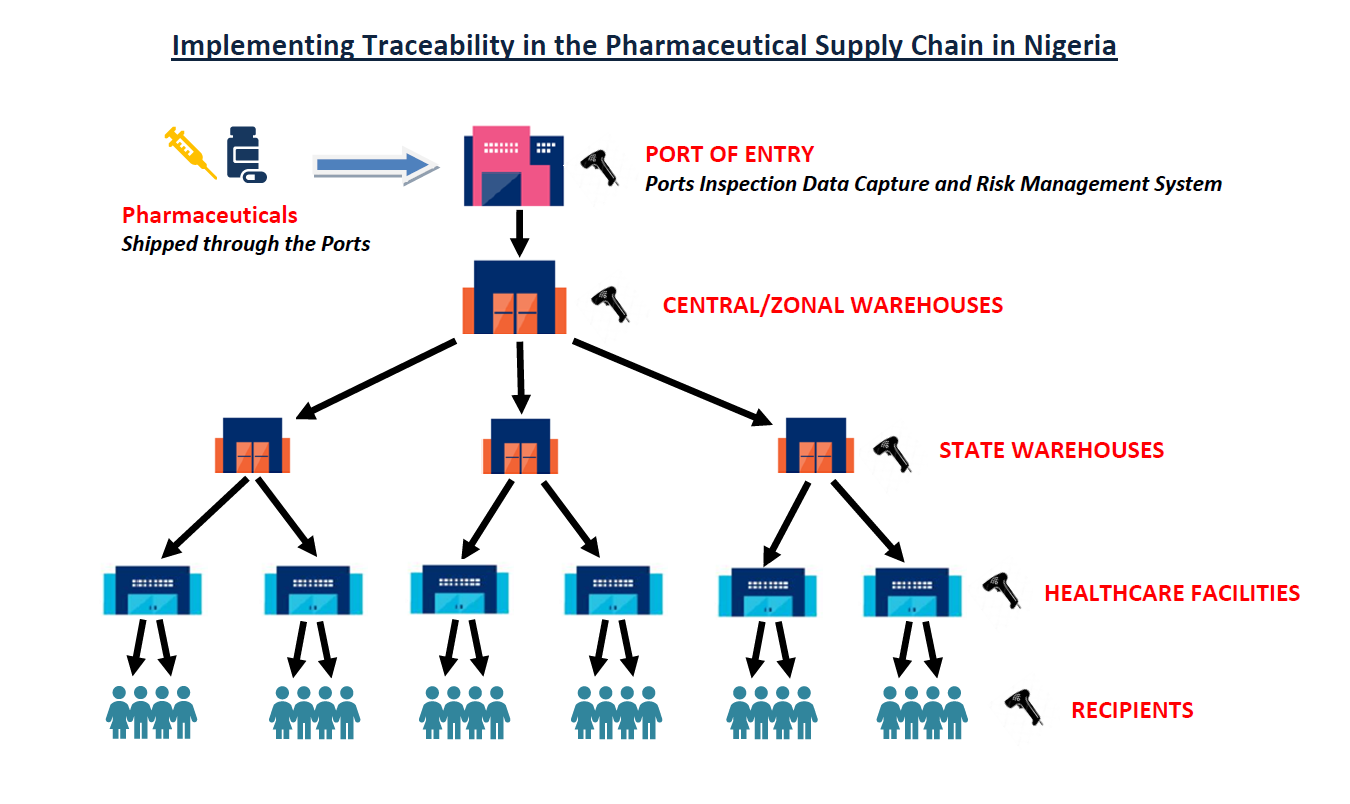
𝐏𝐡𝐚𝐫𝐦𝐚𝐜𝐞𝐮𝐭𝐢𝐜𝐚𝐥 𝐓𝐫𝐚𝐜𝐞𝐚𝐛𝐢𝐥𝐢𝐭𝐲 𝐢𝐧 𝐍𝐢𝐠𝐞𝐫𝐢𝐚 – 𝐒𝐭𝐚𝐭𝐮𝐬 𝐔𝐩𝐝𝐚𝐭𝐞
The regulatory backbone is the National Agency for Food and Drug Administration and Control (NAFDAC) its main objectiv...
𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 𝐌𝐨𝐦𝐞𝐧𝐭𝐮𝐦 𝐢𝐧 𝐀𝐟𝐫𝐜𝐢𝐚
It is fair to state that Track & Trace in Africa has entered a true acceleration phase.
𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 𝐢𝐧 𝐒𝐚𝐮𝐝𝐢 𝐀𝐫𝐚𝐛𝐢𝐚( 𝐅𝐫𝐨𝐦 𝐓𝐫𝐚𝐜𝐞𝐚𝐛𝐢𝐥𝐢𝐭𝐲 𝐭𝐨 𝐕𝐞𝐫𝐢𝐟𝐢𝐜𝐚𝐭𝐢𝐨𝐧)
Let’s shed light on how pharmaceutical traceability evolved in Saudi Arabia under the national platform SFDA - هيئة الغذ...
𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 𝐈𝐦𝐩𝐥𝐞𝐦𝐞𝐧𝐭𝐚𝐭𝐢𝐨𝐧 𝐑𝐨𝐚𝐝𝐦𝐚𝐩 𝐢𝐧 𝐊𝐞𝐧𝐲𝐚
The standard announced in last septmber covers all medicinal, cosmetic, and personal care products consumed, injected, o...
Pre Track & Trace Implementation in Egypt, what you need to do?
With the official start of pharmaceutical track & trace in Egypt scheduled for February for imported medicines, and a tr...
EDA Track & Trace Q&A – Detailed Clarification
these were some Q&A published before in October 2025 by EDA
Warehouses Must Operationally Prepare for SSCC-Based Handling Under EDA’s Decision 804
Under Decision 804, logistics movements between manufacturers, distributors and pharmacies are expected to be reported a...
Substandard and falsified medical products in south Africa
Highlighting some points in the article shared by head of regulatory compliance in SAHPRA
Key Market Insights and Technical Reflections on Egypt’s Track & Trace Draft Guidelines
Reading from EDA announced draft guidelines adn some thought about the Egyptian pharmaceutical market architecture
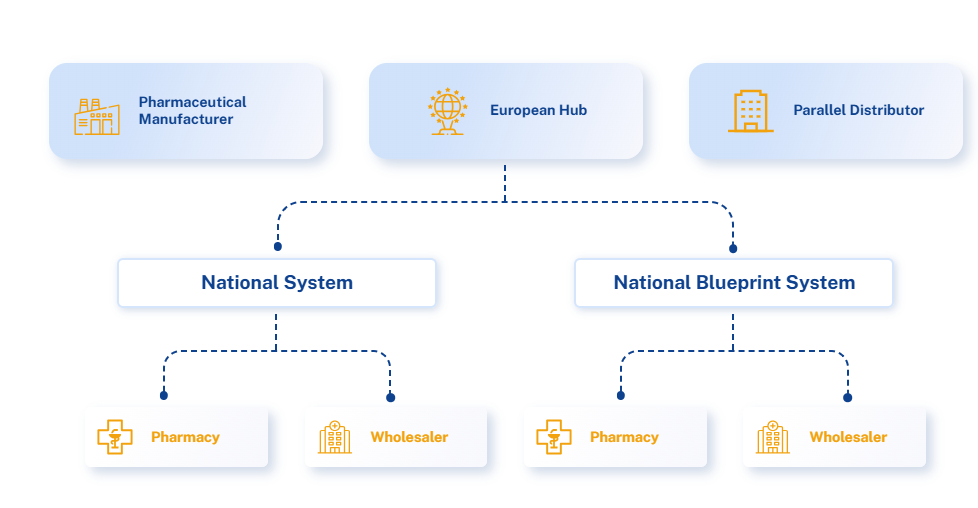
Understanding Egypt’s Pharma Supply Chain
Earlier this month, the Egyptian Drug Authority (EDA) officially announced in the official gazette the establishment of ...
Uzbekistan’s National Hub new requirement
Update in serial generation of items imported to Uzbekistan