Knowledge Base for serialization engineers.
Learn about EPCIS, GS1 Standards, and Track & Trace implementation
A Reading of the Key Differences Between the Draft Regulatory Guide and EDA Law No. 804
There are several fundamental differences between the Draft Regulatory Guide governing the establishment and operation o...
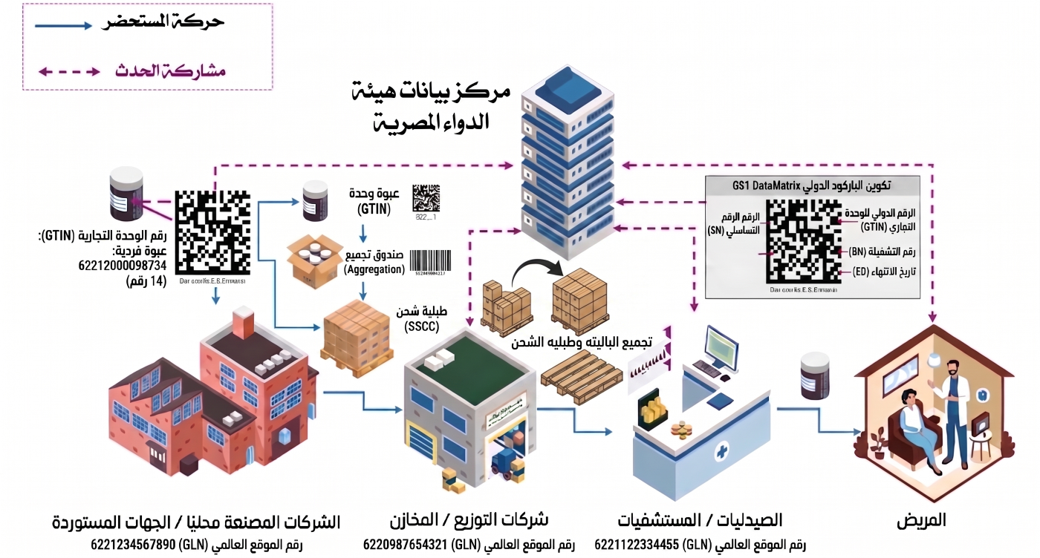
Pharma Supply chain Gaps
How Counterfeit Medicines Slip Into the Supply Chain, And Why It Keeps Happening
𝐖𝐇𝐎 𝐌𝐋𝐬,𝐆𝐁𝐓 𝐚𝐧𝐝 𝐀𝐟𝐫𝐢𝐜𝐚
World Health Organization considers medical products and health technologies one of the six core building blocks of heal...
𝐓𝐫𝐚𝐜𝐞𝐚𝐛𝐢𝐥𝐢𝐭𝐲 𝐒𝐭𝐨𝐫𝐢𝐞𝐬 : 𝐖𝐡𝐲 𝐓𝐫𝐚𝐜𝐞𝐚𝐛𝐢𝐥𝐢𝐭𝐲-Pricing Governance
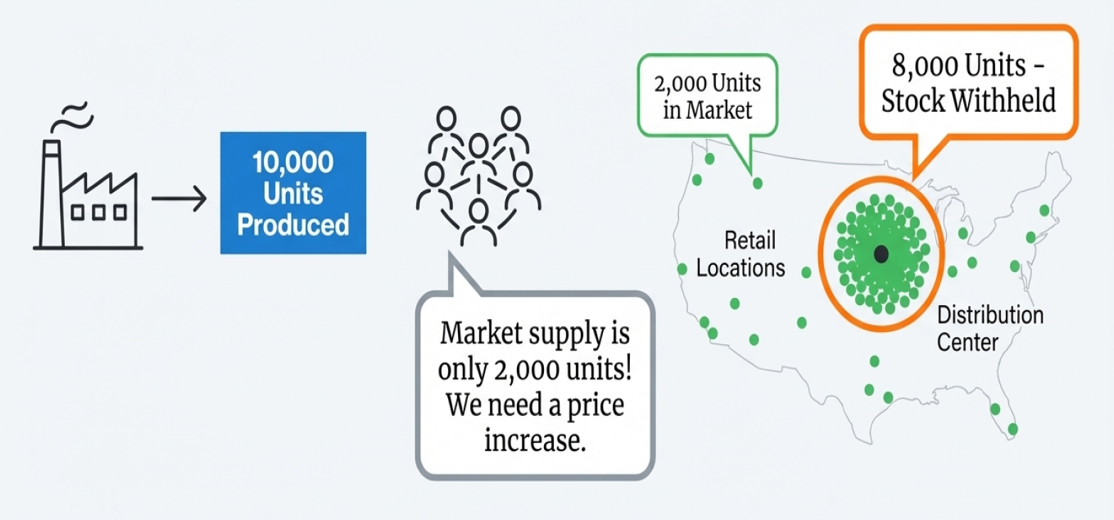
Pricing Pressure, Not Price Hikes: How Data Ends the Illusion of Drug Shortages
Traceability Stories : Why Traceability-Patient Safety
The moment you see the surge of traceability announcements across the Middle East and Africa, one question likely spring...
𝐀𝐮𝐭𝐨𝐦𝐚𝐭𝐢𝐜 𝐈𝐝𝐞𝐧𝐭𝐢𝐟𝐢𝐜𝐚𝐭𝐢𝐨𝐧 & 𝐃𝐚𝐭𝐚 𝐂𝐚𝐩𝐭𝐮𝐫𝐞 (𝐀𝐈𝐃𝐂)
The silent engine behind every serious Track & Trace implementation.
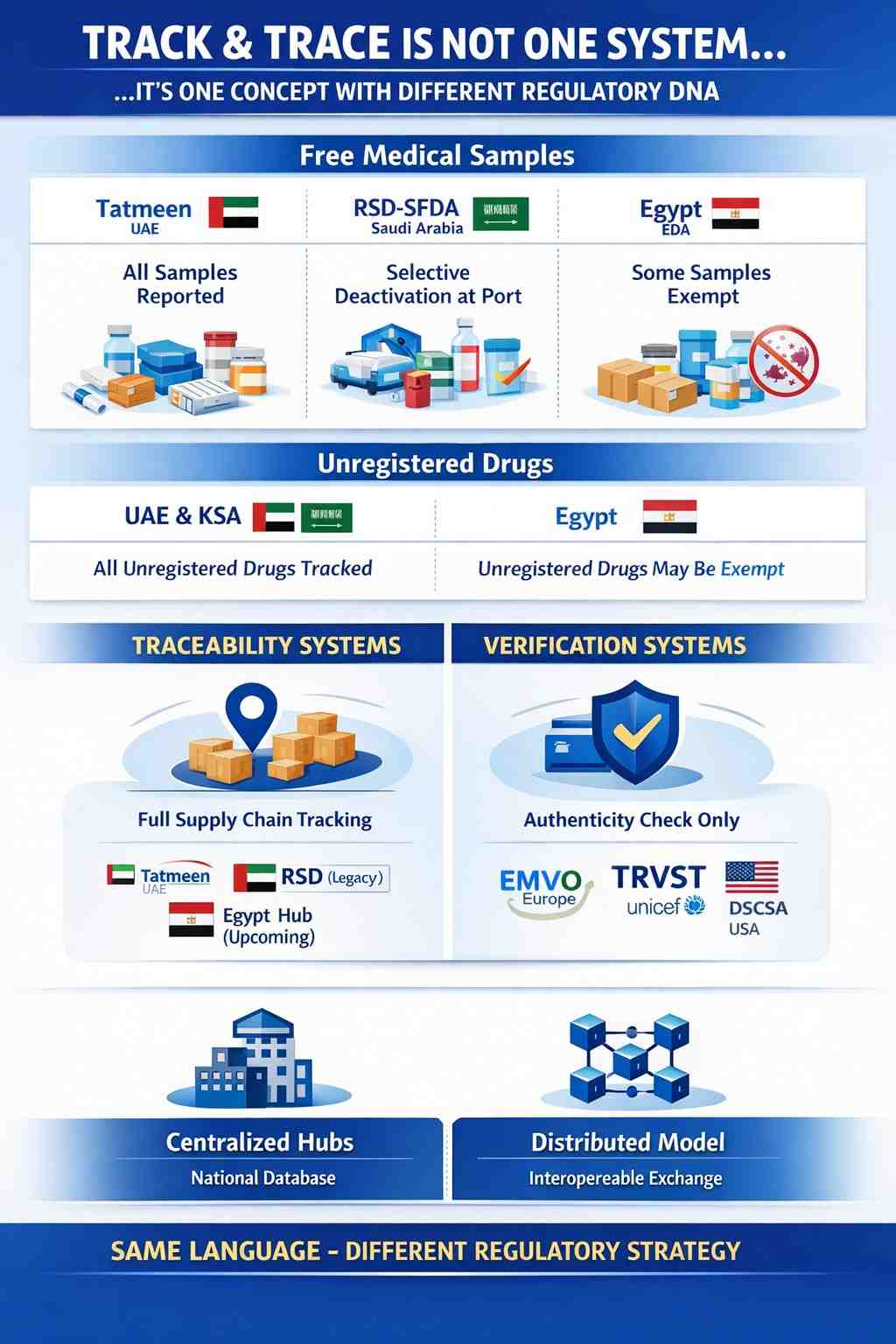
𝐔𝐧𝐩𝐨𝐩𝐮𝐥𝐚𝐫 𝐨𝐩𝐢𝐧𝐢𝐨𝐧: 𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 𝐢𝐬 𝐧𝐨𝐭 𝐨𝐧𝐞 𝐝𝐞𝐬𝐢𝐠𝐧𝐞𝐝 𝐬𝐲𝐬𝐭𝐞𝐦
One of the most common misunderstandings in pharmaceutical serialization is assuming that once you hear “Track & Trace”,...
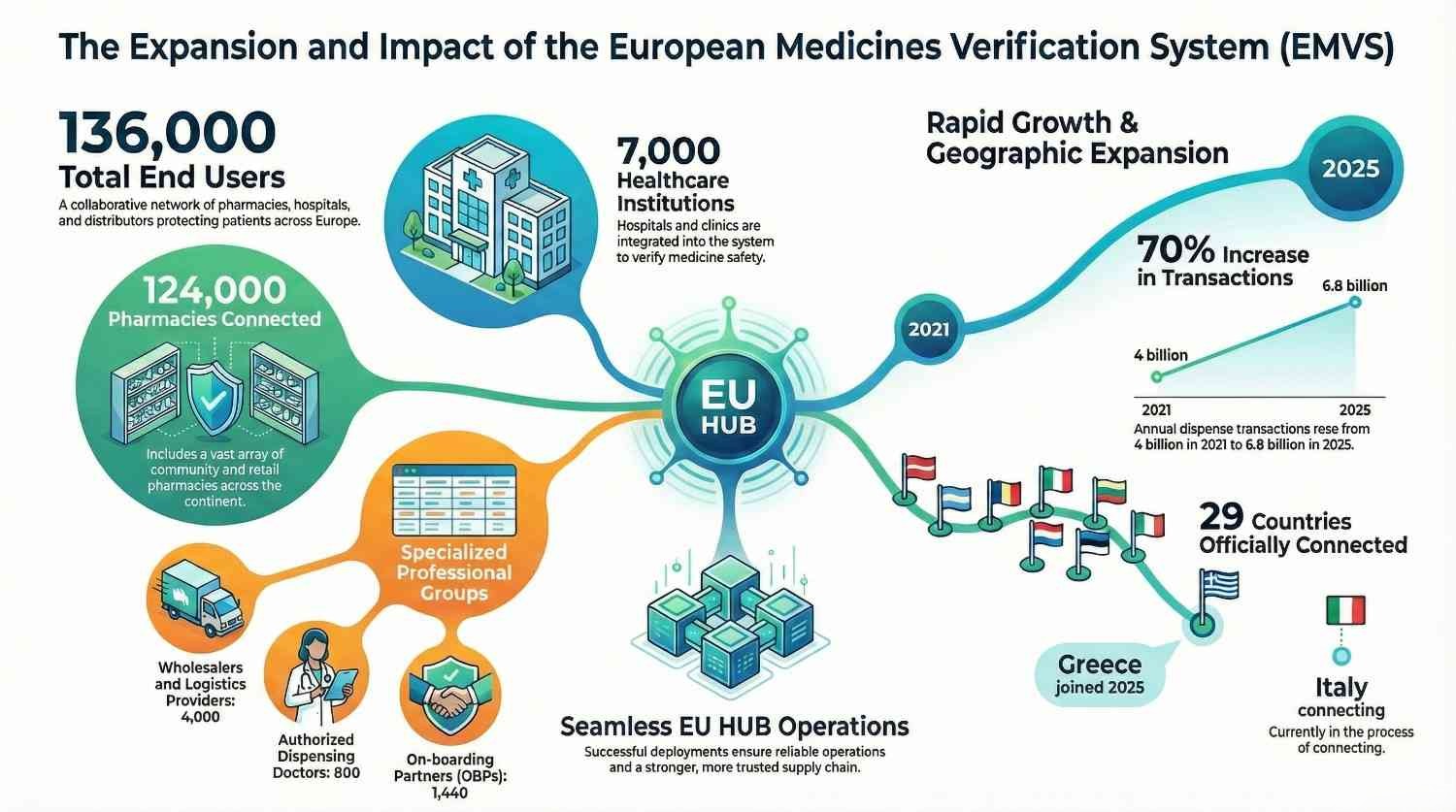
Celebrating 7 years of the European Medicines Verification System (EMVS)
On 9 February 2019, EMVS went live. On 9 February 2026,marks seven years of continuous operation securing the pharmaceu...
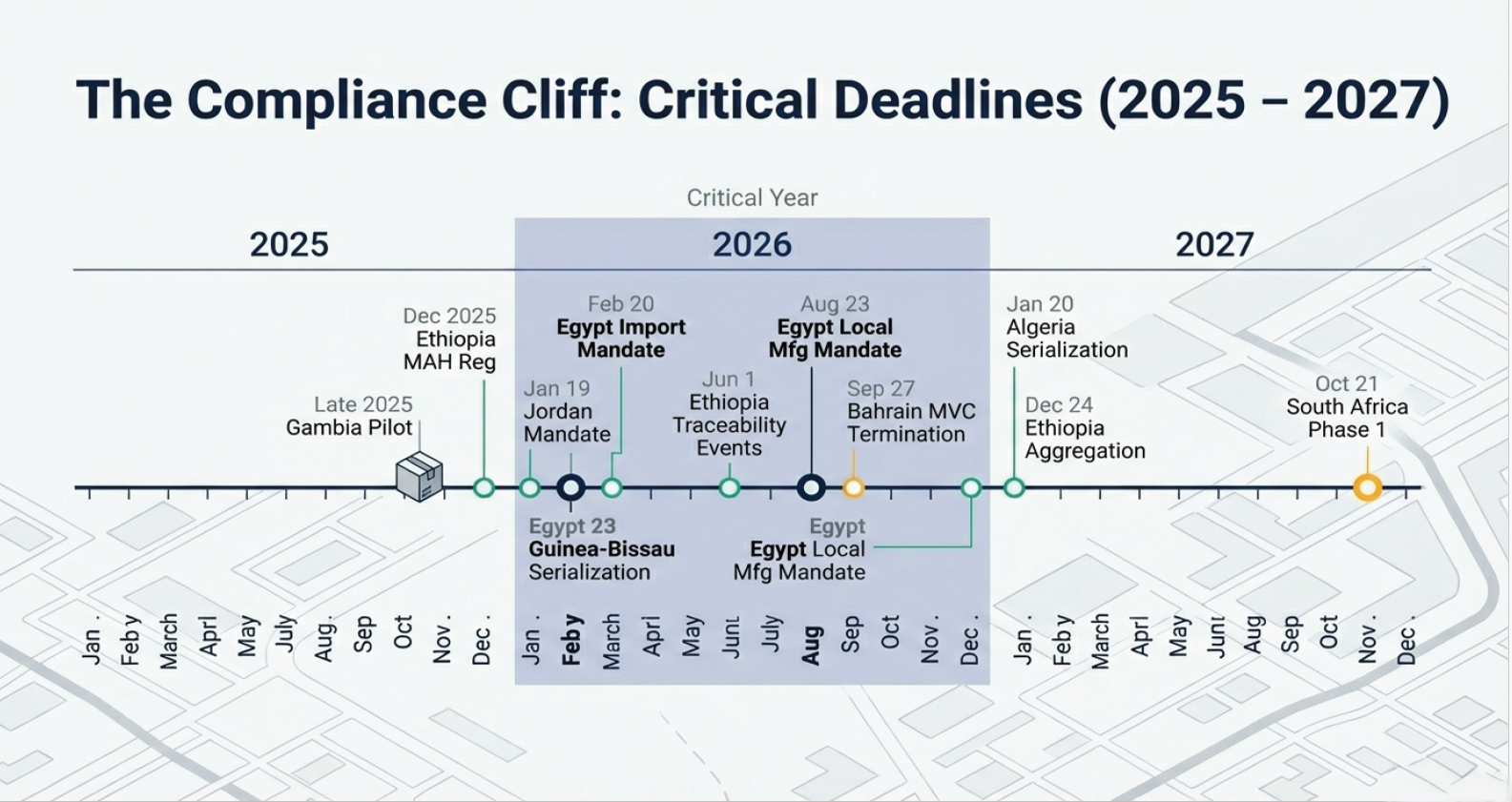
𝟐𝟎𝟐𝟔 𝐑𝐨𝐚𝐝𝐦𝐚𝐩 𝐟𝐨𝐫 𝐌𝐄𝐀 𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞
This year marks a historic surge in pharmaceutical traceability across Africa and the Middle East. For Marketing Authori...
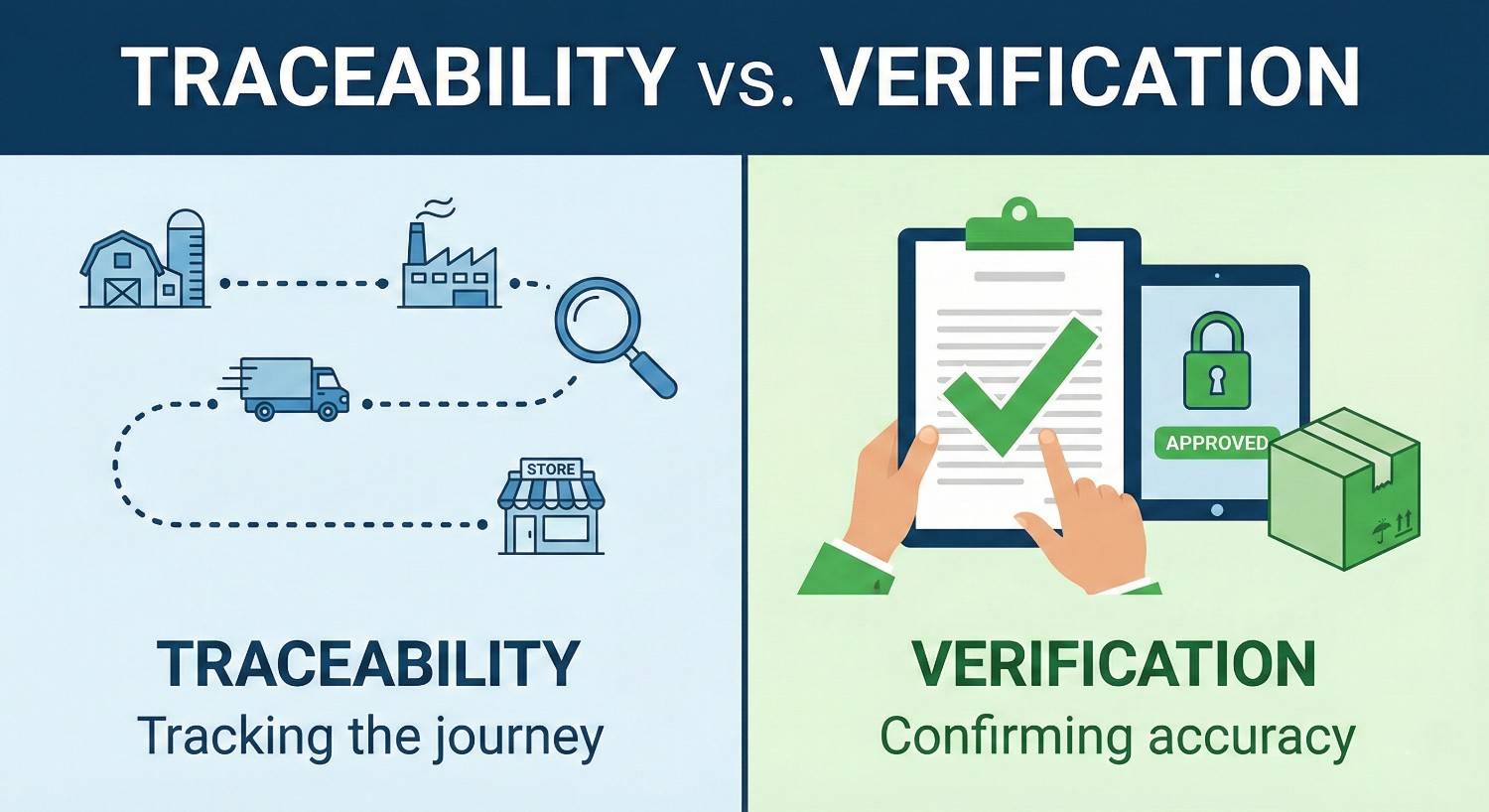
Traceability vs Verification
There is a common misconception around some key expressions related to Track & Trace, and this really needs clarificatio...
Mobile Application in Track and Trace Reporting
Most Track & Trace hubs rely on traditional reporting channels: APIs for system-to-system integration, and sometime...
GS1 Data Matrix Exceptions
If you work across the Middle East and Africa, most serialization guidelines are straightforward.
Price governance from Reactive to Proactive
Implementing Track and trace and through serialization allows regulatory bodies to transition from reactive pricing mode...
How Traceability can serve to enhance Pharmacovigilance
Rethinking Point of view beyond compliance
The Hidden Cost of Choosing the Wrong Track & Trace Software
Selecting the wrong way to report the track & trace events can be one of the most expensive mistakes a company makes.
Is it time to rethink e-leaflets in alignment with pharmaceutical serialization?
Rethinking Point of view beyond compliance
𝐖𝐡𝐞𝐧 𝐓𝐫𝐚𝐜𝐤 & 𝐓𝐫𝐚𝐜𝐞 𝐌𝐞𝐞𝐭𝐬 𝐋𝐞𝐠𝐚𝐜𝐲 𝐒𝐲𝐬𝐭𝐞𝐦𝐬 (𝐌𝐨𝐦𝐞𝐧𝐭 𝐎𝐟 𝐓𝐫𝐮𝐭𝐡)
As countries across the region push forward with national serialization mandates (like Tatmeen in UAE, RSD in KSA, and N...
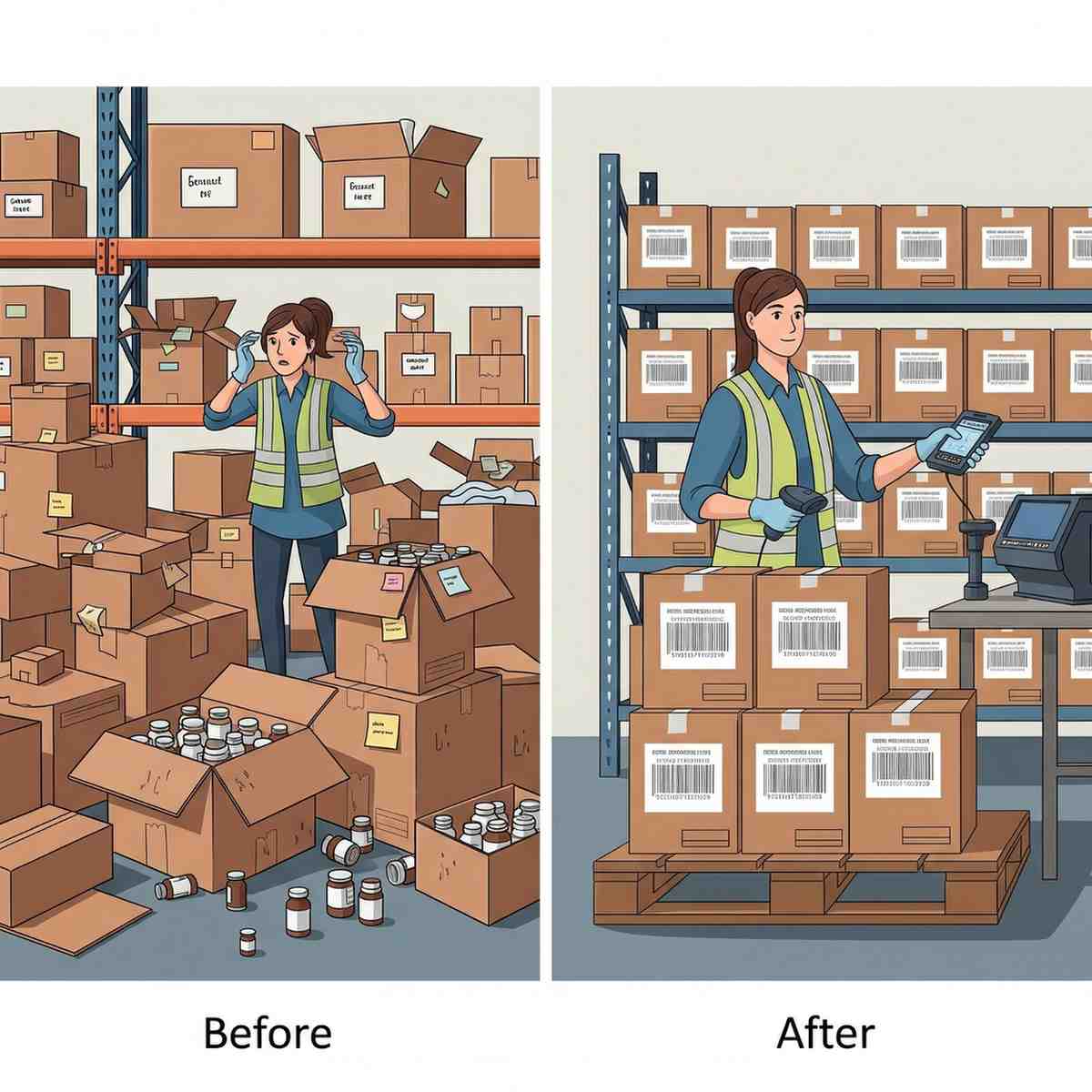
Serialization Impact on warehouse performance
Serialization is a game-changer for warehouse management. In this era where precision and traceability taking place in m...
Pharmaceutical Track & Trace Momentum Across Middle East & Africa
In just the last two quarters of 2025, nearly 10 countries across the Middle East and Africa have announced launching th...
The Weak Links in the Global Pharma Supply Chain
Insights from WHO, Pharmaceutical Security Institute (PSI) Situation Reports.
When Your Current WMS / HIS / POS / ERP Meets Track & Trace: Adapt or Struggle
The Track & Trace journey has reached a major milestone , it’s already live in KSA, UAE, and Bahrain, with rollouts star...
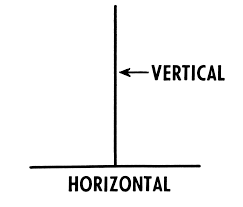
Track vs Trace in pharmaceuticals
Track: Monitoring the current location of products in real-time. Tr...
Aggregation as a Service (AaaS)
Pharma manufacturers today face a tough decision when it comes to track & trace compliance